The new NMR technology reported by the Gossert group in "Angewandte Chemie Intl. Ed." enables studying proteins >100 kDa without the need of complicated deuteration protocols. This facilitates NMR studies of proteins produced in mammalian cells like GPCRs, therapeutic antibodies and large molecular complexes.
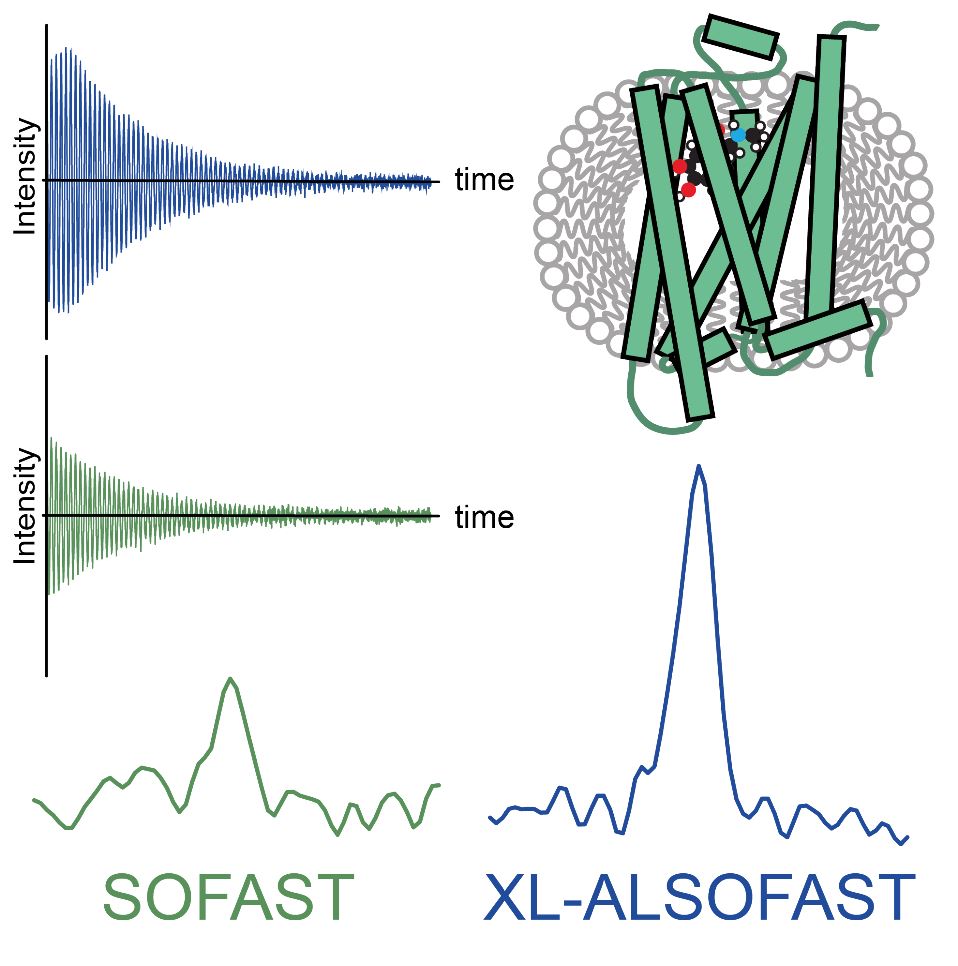
Conventional NMR suffers from a size limitation, and biomolecules >30 kDa can only be studied using advanced deuterium labeling protocols. The new method introduced by Rößler et al. allows obtaining highly sensitive NMR spectra of particles >100 kDa, without the need for deuteration. Thus, also proteins produced in eukaryotic cells, where deuteration is hardly feasible, can be studied by NMR.
The applications described in the work of the Gossert group include first examples of functional studies of GPCRs expressed in HEK cells. Here, a spectrum can now be recorded in less than an hour compared to nearly a day with conventional methods, allowing studies of unstable complexes. Further, high-quality NMR spectra of large molecular assemblies of up to 240 kDa are shown. The probably most widespread application will be in the pharmaceutical industry, where with the new experiment therapeutic antibodies of 150 kDa can be analyzed directly from the injection vial without further modification.
This newly established technique will therefore expand the range of biomolecular systems that are accessible to NMR studies at the atomic level.
Link to the paper in "Angewandte Chemie Intl. Ed."






