Hydrogen is a good energy vector that can be sustainably produced through water electrolysis - but its production is slowed down by the low efficiency of the oxygen evolution reaction. Iridium oxide is the material of choice today for water electrolysis under acidic conditions, but it is a very scarce metal, hence the need to minimize its quantity. Christophe Copéret and his team developed an efficient catalyst based on single iridium atoms dispersed on a conductive indium tin oxide support.
Entering Copéret's laboratories, one gets his/her attention caught by black rubber hands protruding from glove-boxes, where it is possible to prepare catalysts and carry out reactions under the most pristine and controllable conditions. One of the research activities of the Copéret group focuses on water electrolysis, a reaction where water is split into hydrogen and oxygen via electric current - a method that is mainly used to obtain hydrogen for energy storage. However, its production from fossil energy sources (mostly methane) is still cheaper. One reason for this is that the electrolysis of water, especially water oxidation, is extremely complex and not yet understood in all details. The group of Christophe Copéret has now succeeded in developing an efficient catalyst for the electrochemical splitting of water and has gained important insights into the reaction mechanism.

The higher up you start, the easier you reach the top
The catalytic splitting of water consists of two half-reactions: the release of hydrogen (Hydrogen Evolution Reaction, HER) and the release of oxygen (Oxygen Evolution Reaction, OER), but the latter is the more complex one. The search for stable and cheaper catalysts for the OER and the precise understanding of the reaction mechanism remain of great importance. "You can imagine this reaction like high-altitude mountaineering and a Mont-Blanc excursion," Copéret explains. This "mountain chain" must be reached for the reaction to take place. "Electrochemistry shortens the distance and acts as an 'elevator' up to the first 'summit station'. The catalyst, which is supposed to further accelerate the reaction, makes the trip through the various mountain passes even easier - provided you choose the right material. If you want to work at such an elevation (= the potential in chemistry), you must be fit and resistant and that also applies to the catalyst. We refer to this as the activity and stability, respectively".
From water to oxygen on a single iridium atom
The Copéret group uses the heterogeneous form of catalysis for water electrolysis (solid electrocatalysts). This means that the catalyst and the reaction partners are present in different phases making easy the separation of products - this solid catalyst is surrounded by the liquid reaction partner (water) and plays the role of an electrode on which a potential is applied to drive the reaction. The environment also plays an important role. The half-reactions of water electrolysis can take place in an acidic or alkaline environment. The reaction in an acidic environment offers some advantages - so it is the preferred one - but it is also strongly limited by the catalyst stability and cost. Currently, the most robust catalysts for water oxidation (OER) under acidic conditions is iridium oxide, a very rare and expensive metal.
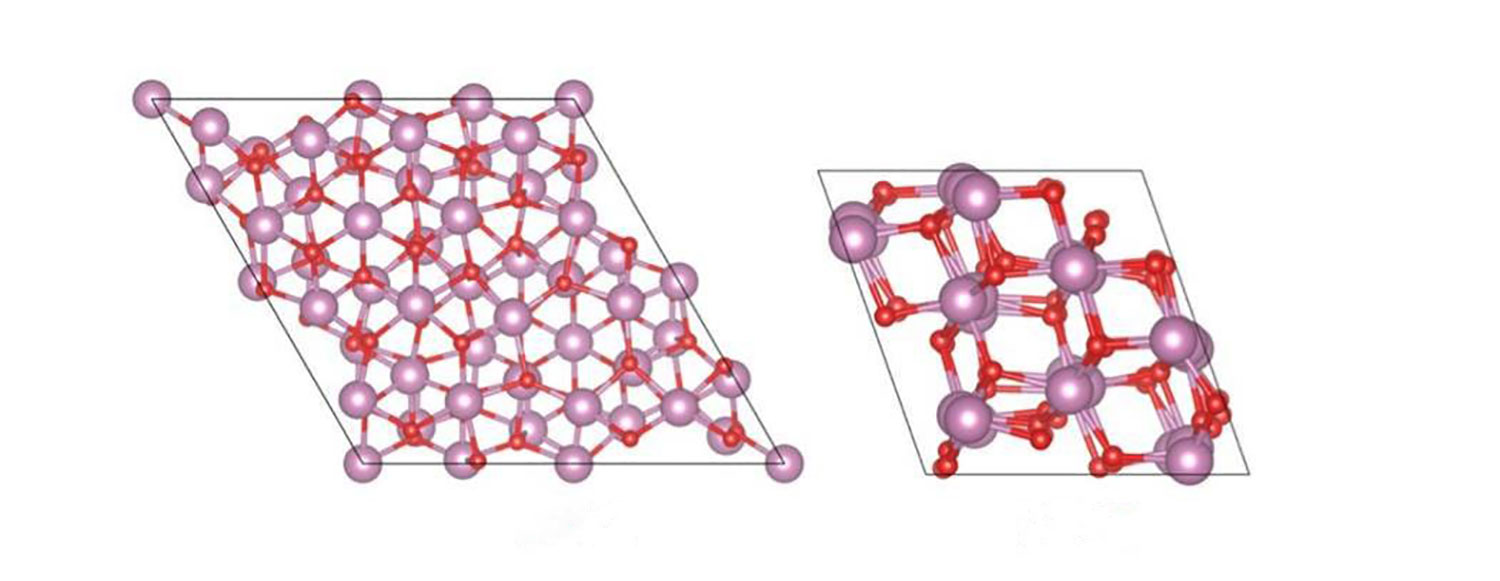
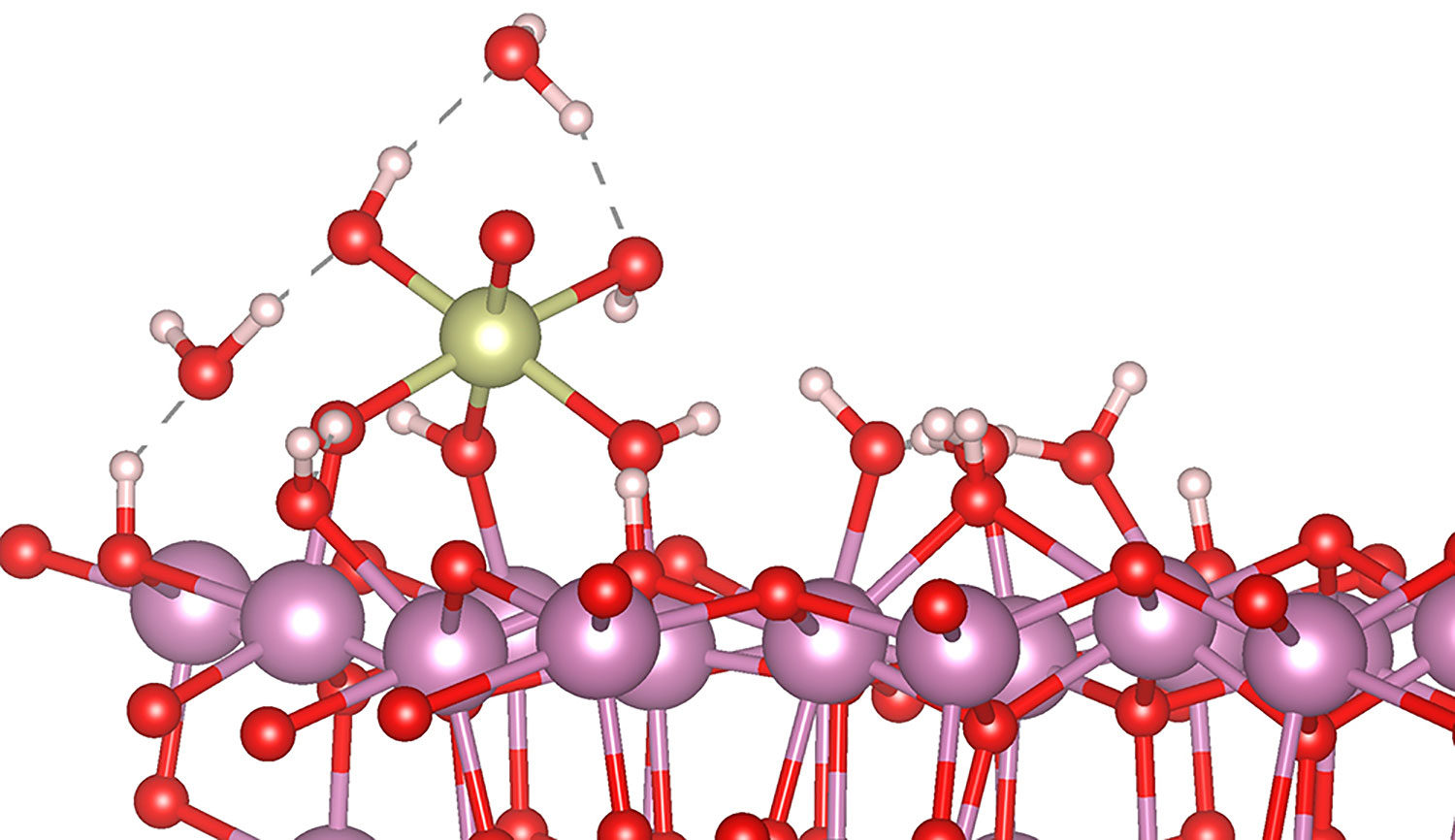
To reduce the amount of iridium to a minimum, Copéret and his team have chosen to fix individual iridium atoms at the surface of a conductive Indium tin oxide (ITO) support. This system constitutes the first reported and fully characterized iridium single atom catalyst (SAC) stable under acidic conditions according to Copéret. In addition to reducing the necessary amount of the expensive, rare iridium as much as possible, this approach ensures that every atom is involved in catalysis and that detailed mechanistic studies can be undertaken (all spectroscopic investigations interrogate the active sites). ITO was used because the usual carbon-based support materials are not stable enough under these reaction conditions.
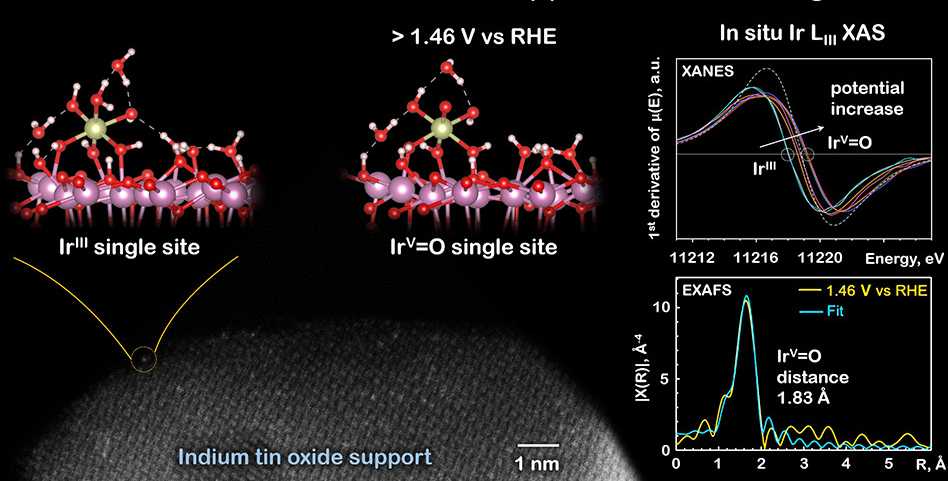
Using electrochemical, spectroscopic and microscopic methods, the group was able to show that the iridium atoms on the support were really single ones, each generating hundreds of oxygen molecules during the catalytic tests. The in-situ X-Ray absorption experiments revealed details of the reaction process in the acidic environment and allow detecting an iridium(V) oxo intermediate. By modelling the reaction mechanism, the team was able to show that the observed iridium(V) oxo intermediate (IrV-O distance of 1.83 Å) is probably a catalytic resting state. The intermediate is further oxidized to IrVI, which enables a fast attack of the nucleophilic water molecule. The results further illustrate that SACs on oxide surfaces are applicable to a broad range of heterogeneously catalyzed transformations.
About Methanol Economy & ACS Live Presentations
"Of course, this result alone does not solve the energy problem," Copéret says "but the results can help to design better materials and processes." In addition, the use of transparent electrodes could also make this catalyst amiable photo-electrochemical process, such as solar driven water splitting. Copéret also elaborates: "If we have access to H2 from a renewable source such as water electrolysis, one could further react it with CO2 to form methanol, a liquid fuel. This is the basic concept of the "methanol economy" that could help to close the carbon cycle and mitigate climate change." There are still plenty of challenges to meet the targets, but this iridium project - a project in part sponsored by the InnoSuisse project (SCCER Heat and Energy Storage) - is a step forward in water electrolysis, as can be seen as well in the team's live presentation on their ACS paper. Combination of these research fields is currently ongoing in the group.