
University of Toronto physicians say more research is needed on the popular antiviral drug remdesivir, after they documented a rare case of bradycardia in a male COVID-19 patient shortly after he received the medication.
Bradycardia, or an abnormally slow heart rate, is when the heart beats less than 60 times per minute. It can be life-threatening if the heart is unable to maintain a rate that pumps enough oxygen-rich blood throughout the body.
The case study, published in the Canadian Medical Association Journal, highlights a potential rare and serious association between remdesivir therapy and bradycardia observed in a Sinai Health patient. It involved the Temerty Faculty of Medicine's Leora Branfield Day, a resident in the department of medicine's general internal medicine program, and Michael Fralick, a clinician scientist with the department of medicine and a general internist at Sinai Health.
Day, a fifth-year resident, said researchers noted the condition in one of their hospitalized COVID-19 patients soon after starting remdesivir treatment.
The patient, who was 59 and otherwise healthy and had no other risk factors for bradycardia, later recovered. However, the bradycardia only went away when they stopped the medication, Day noted.
"Given the frequent use of remdesivir in the treatment of patients hospitalized with COVID-19, clinicians should be aware of the potential risk of bradycardia, particularly when treating patients at elevated baseline risk," said Day.
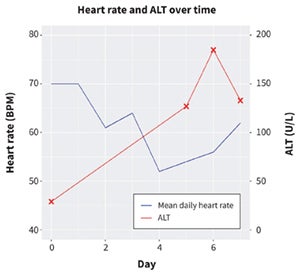
Changes in mean daily heart rate (beats per minute [BPM]) and alanine aminotransferase (ALT; units/L) from day of admission to discharge. Remdesivir was started on day two and discontinued on day five.
Remdesivir has been shown to reduce the length of time moderately ill COVID-19 patients spend in hospital. It is approved by Health Canada for the treatment of hospitalized patients with COVID-19 pneumonia who require oxygen.
Globally, it is one of the most used medications to treat patients hospitalized with COVID-19, with more than two million doses estimated to have been administered worldwide during the pandemic.
"The bradycardia observed in our patient was suggestive of a probable adverse drug reaction," said Day, adding there have been other similar cases recently described with the use of remdesivir in COVID-19 patients. "This reinforces the need for further studies to explore whether this might be a rare drug reaction in this medication used frequently for patients with COVID-19 pneumonia."
Risk factors for bradycardia include increasing age, cardiovascular disease, and the use of medications that can cause bradycardia, including beta blockers.






