Scientists at Tokyo Institute of Technology (Tokyo Tech), Imperial and High Energy Accelerator Research Organization (KEK) Institute of Materials Structure Science, discover new Ba7Nb4MoO20-based materials with high oxygen-ion (oxide-ion O2-) conductivities—"the hexagonal perovskite-related oxides"—and shed light on the underlying mechanisms responsible for their conductivity. Their findings lead the way to uncovering other similar materials, furthering research on developing low-cost and scalable renewable energy technologies.
Over the past few years, fuel cells have become a focal point of research in eco-friendly technology because of their superior abilities to store and produce renewable energy and clean fuel. A typical type of fuel cell gaining ground is the oxide-ion-conducting fuel cell, which is primarily made of materials through which oxide ions (oxygen ions: O2-), can easily move. New materials with higher conductivity at low and intermediate temperatures, provide a number of advantages over commonly used fuel cells based on yttria-stabilized zirconia (YSZ) electrolytes, such as higher power generation efficiency, longer lifetimes, and lower costs.
However, only a limited number of such materials are known and their application to developing fuel cells has largely remained at the laboratory scale. To truly achieve a sustainable energy economy, new oxide-ion conductors with high conductivity need to be discovered that can allow low-cost and efficient scaling up of these technologies.
Scientists from Tokyo Tech, Imperial and KEK set out to address this need, and in a recent study, identified a new oxide-ion-conducting material that may be a representative of an entire family of oxide-ion conductors.
The material in question has the chemical formula Ba7Nb3.9Mo1.1O20.05 and is classified as a "hexagonal perovskite-related oxide." Prof Masatomo Yashima, who led the study, explains: "Ba7Nb3.9Mo1.1O20.05 shows a wide stability range and predominantly oxide-ion conduction in the oxygen partial pressure range from 2 × 10-26 to 1 atm. Surprisingly, bulk conductivity of Ba7Nb3.9Mo1.1O20.05, 5.8 × 10-4 S/cm, is remarkably high at 310 oC, and higher than bismuth oxide- and zirconia-based materials (Fig. 1)."* Prof Stephen Skinner comments that the fast oxide ion transport was unambiguously confirmed using the 18O tracer diffusion technique at Imperial.
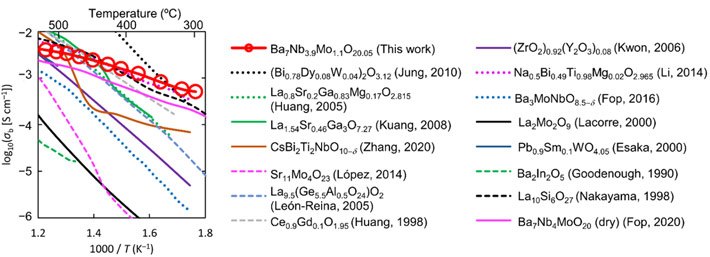
Figure 1. Comparison of bulk conductivities σb of Ba7Nb3.9Mo1.1O20.05 and other oxide-ion conductors. *
Prof Yashima and his team note that the crystal structure of Ba7Nb3.9Mo1.1O20.05 contains oxygen-deficient layers (c' in Fig. 2a and Fig. 2c) and that its high oxide-ion conductivity is attributable to the oxide-ion migration on the c' layers. In fact, they succeed in experimental visualization of O1−O5 oxide-ion diffusion pathways by the neutron-diffraction measurements at a high temperature 800 oC with SuperHRPD diffractometer of Prof Takashi Kamiyama's group at KEK/J-PARC (Figs. 2b,d). Prof Yashima says that the oxide ions migrate via interstitialcy diffusion mechanism through interstitial octahedral O5 and lattice tetrahedral O1 oxygen sites and that the (tetrahedral)–(octahedral) diffusion pathways on the c' layer in Ba7Nb3.9Mo1.1O20.05 is the same as those in another hexagonal perovskite-related oxide Ba3MoNbO8.5–δ. Therefore, Prof Yashima and his team claim that "The common feature of the diffusion mechanism would be a guide for design of oxide-ion conductors with the hexagonal perovskite related structures and that the present finding of high oxide-ion conductivities in rare-earth-free Ba7Nb3.9Mo1.1O20.05 suggests the ability of various hexagonal perovskite related oxides as superior oxide-ion conductors".*
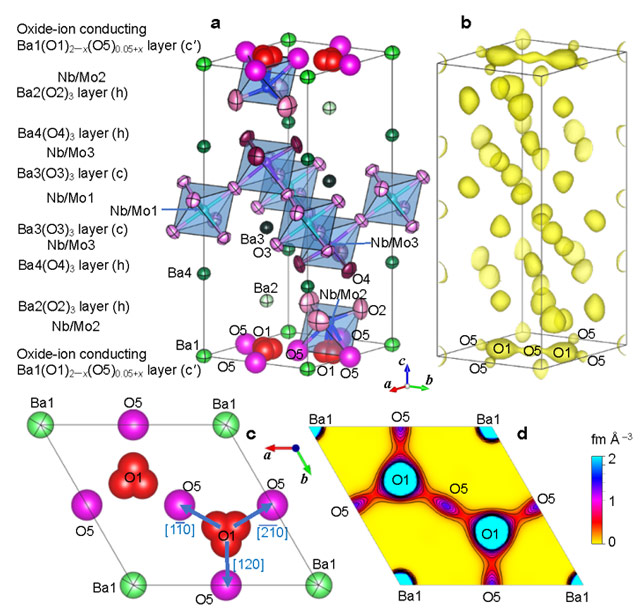
Figure 2. Experimental evidence of the interstitial oxygen O5 and the O1−O5 oxide-ion interstitialcy diffusion of Ba7Nb3.9Mo1.1O20.05 at a high temperature of 800 oC.
(a) Refined crystal structure and (b) corresponding yellow isosurface of maximum-entropy method neutron scattering length densities (MEM NSLDs) at 0.36 fm/Å3 of Ba7Nb3.9Mo1.1O20.05 at 800 oC. Refined crystal structure (c) and corresponding MEM NSLD distribution (d) on the ab plane at z = 0 of Ba7Nb3.9Mo1.1O20.05 at 800 oC. In panel d, the contour lines from 0 to 2 fm/Å3 by the step of 0.2 fm/Å3. Arrows in panel c denote the directions of oxide-ion O1-to-O5 migration.






