Scientists at Tokyo Institute of Technology (Tokyo Tech) found a way to synthesize a special type of perovskite that promotes the production of ammonia, which has key applications in fertilizer production and hydrogen energy. This new synthesis process can be carried out at temperatures immensely lower than that of traditional methods for the synthesis of perovskite-oxide materials and in much less time, and the produced perovskite outperforms all its competitors for producing ammonia.
Perovskites are a class of synthetic materials that have a crystalline structure similar to that of the naturally occurring mineral calcium titanate. They have been the subject of many studies because they exhibit exciting and unique properties that can be tuned according to their composition. One of their potential applications is as catalysts for the synthesis of ammonia. In other words, specific perovskites can be placed inside a reaction chamber with nitrogen and hydrogen to promote the reaction of these gases to form ammonia.
Ammonia is a useful substance that can be employed in the production of fertilizers and artificial chemicals, and even as a clean-energy carrier (in the form of hydrogen), which may be key in eco-friendly technologies. However, there are various challenges associated with the synthesis of ammonia and perovskites themselves.
The synthesis rate for ammonia is generally limited by the high energy required to dissociate nitrogen molecules. Some researchers have had some success using precious metals, such as ruthenium. Recently, perovskites with some of their oxygen atoms replaced by hydrogen and nitrogen ions have been developed as efficient catalysts for ammonia synthesis. However, the traditional synthesis of perovskites with such substitutions usually has to be carried out at high temperatures (more than 800 ℃) and over long periods of time (weeks).
To address these issues, in a recent study carried out at Tokyo Tech, a group of researchers led by Prof. Masaaki Kitano devised a novel method for the low-temperature synthesis of one of such oxygen-substituted perovskites with the chemical name BaCeO3−xNyHz and tested its performance as a catalyst to produce ammonia. To achieve this, they made an innovative alteration to the perovskite synthesis process. The use of Barium carbonate and Cerium dioxide as precursors (or "ingredients") involves a very high temperature would be required to have them combine into the base perovskite, or BaCeO3, because Barium carbonate is very stable. In addition, one would then have to substitute the oxygen atoms with nitrogen and hydrogen ions. On the other hand, the team found that the compound Barium amide reacts easily with Cerium dioxide under ammonia gas flow to directly form BaCeO3−xNyHz at low temperatures and in less time (Fig. 1). "This is the first demonstration of a bottom-up synthesis of such a material, referred to as perovskite-type oxynitride-hydride," explains Prof. Kitano.
Finally, the researchers attempted to elucidate the mechanisms behind the improved synthesis rate for ammonia. Overall, the insight provided in this study serves as a protocol for the synthesis of other types of materials with nitrogen and hydrogen ion substitutions and for the intelligent design of catalysts. "Our results will pave the way in new catalyst design strategies for low-temperature ammonia synthesis," concludes Prof. Kitano. These findings will hopefully make the synthesis of useful materials cleaner and more energy efficient.
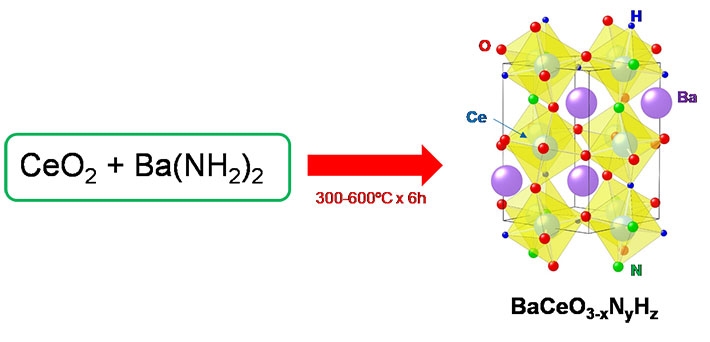
Figure 1. Synthesis of the proposed perovskite
This new protocol for the production of BaCeO3−xNyHz can be carried out at much lower temperatures and in much less time compared with conventional methods.
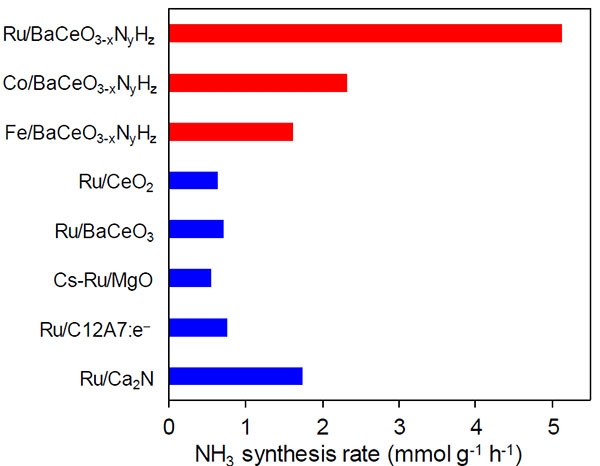
Figure 2. Synthesis rates for ammonia of various catalysts
The proposed perovskite is a much better catalyst than other state-of-the-art materials, especially when combined with more common metals, such as cobalt or iron.






