Opening new possibilities for fluorescence-based imaging in extreme environments, from bioimaging to advanced materials

Since the 1960s, boron-dipyrromethene dyes, commonly called BODIPY dyes, have been widely used for their strong fluorescence, especially in bioimaging, molecular and ion sensing, and as photosensitizers. Researchers especially like how, with simple modifications to BODIPY molecules, their emission color can be tuned-an indispensable quality for multicolor imaging applications.
However, conventional BODIPY dyes are unstable in acidic environments. Strong acids can disrupt their structure by removing the boron atom and causing the dye to lose its fluorescence. This has limited their use in highly acidic conditions. In a new breakthrough, researchers from Hokkaido University have developed a superacid-resistant BODIPY dye. The research team, led by Professor Yasuhide Inokuma at the Institute for Chemical Reaction Design and Discovery (WPI-ICReDD), reports its findings in Nature Communications on March 19, 2026.
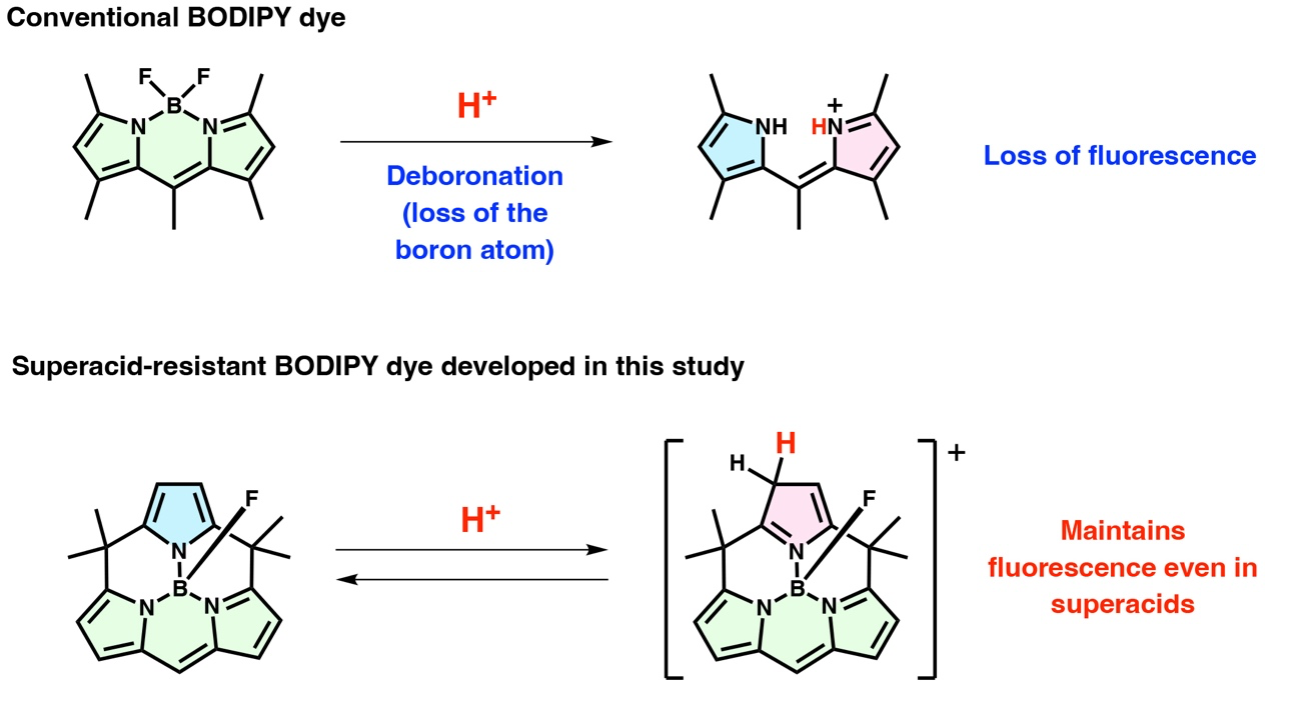
Conventional BODIPY dyes consist of a boron atom bonded to two pyrrole rings. In comparison, the new dye has a third pyrrole ring bonded to the boron atom. This modification introduces an unusual "on/off" fluorescence mechanism. Under normal conditions, the additional pyrrole ring suppresses fluorescence, a process known as quenching. But, in the presence of strong acids, this third ring becomes protonated, stopping the quenching effect and switching the fluorescence on.
The team tested the performance of the new dye in extremely acidic environments. When they used fluorosulfonic acid, which is 1000× stronger than sulfuric acid, the new dye maintained stable fluorescence for more than a day without breaking down. By comparison, even the most acid-resistant BODIPY dyes previously reported could only fluoresce for a few minutes under similar conditions before decomposing.
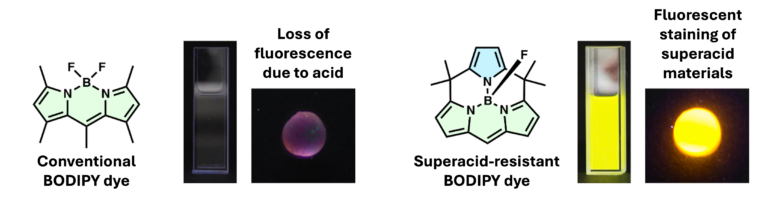
The dye also proved effective for fluorescent staining of strongly acidic materials, including Nafion® beads, sulfonated gels, and ion-exchange resins-materials that are typically difficult to study using conventional BODIPY dyes. It could lead to new analytical techniques for visualizing processes like material degradation and changes in acidity.
The research opens exciting avenues for fluorescence-based imaging and sensor applications in extreme environments involving strong acids and superacids.

Original article:
Keita Watanabe et al. Super-acid resistant macrocyclic BODIPYs. Nature Communications. March 19, 2026. DOI: 10.1038/s41467-026-70499-9
Funding:
This study was partly supported by a JSPS Grant-in-Aid for Challenging Research (Exploratory) (No. JP24K2178704), a JST FOREST Program (No. JPMJFR211H) and the Asahi Glass Foundation, of which Y. Inokuma is the principal investigator. This work was supported by a JSPS Grant-In-Aid for Early-Career Scientists (grant no. 25K18056) to Y. Ide, and the NorthTec Foundation and grant-in-aid for scientific research (grant no. 25K08625) to T. Y. This work was supported by the Institute for Chemical Reaction Design and Discovery (ICReDD), which was established by the World Premier International Research Initiative (WPI), MEXT, Japan.






