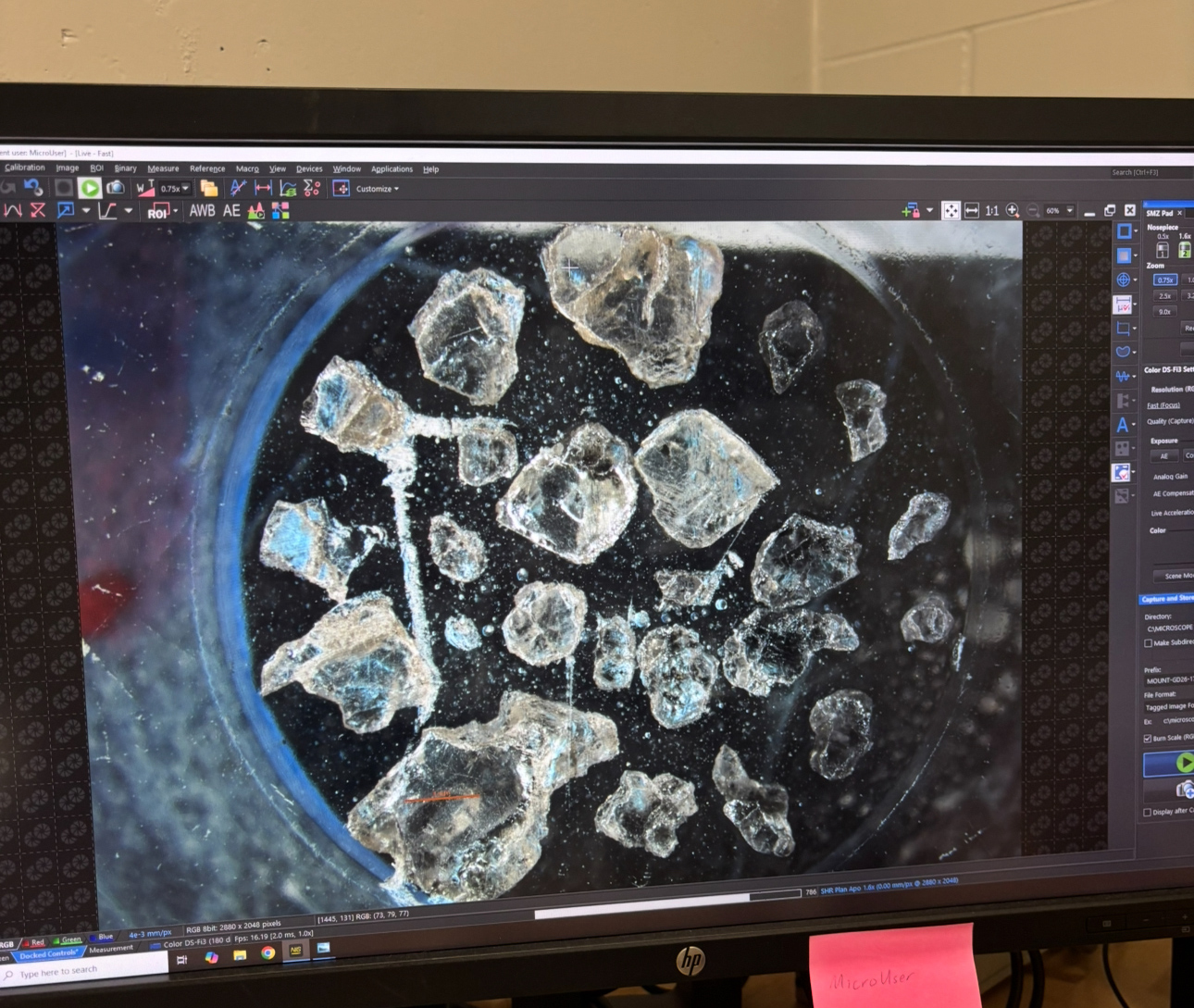
Diamonds are the hardest mineral on Earth, useful in industry and sought after for their beauty. But for scientists, they also offer a window into the deep underground, giving us a glimpse into the mysterious processes that create and shape rock.
Binghamton University senior Kate Halpin had the opportunity to study diamond geochemistry this summer during an internship at Carnegie Science in Washington, D.C.
A love for the outdoors initially sparked the Long Island native's curiosity about the world around her and the unseen processes that shape landscapes and rock formations. During her first year at Binghamton, she took a class with Associate Professor Alex Nikulin on earthquakes and volcanoes that proved transformative.
"I realized that it was something I wanted to pursue. The more I learn about geology, the more I'm curious about it," explained Halpin, a geology major.

Looking for an internship to help prepare for graduate school, Halpin discovered a list of Research Experience for Undergraduates (REU); Carnegie Science was considered one of the best. She spent 10 weeks in the nation's capital, investigating a geological paradox: The composition of a particular set of rocks from southern Africa wasn't considered favorable for the formation of diamonds. Why, then, were diamonds scattered through them like raisins in bread?
The rocks in question come from the Liqhobong kimberlite pipe in Lesotho, Africa, formed by explosive volcanic eruptions that bring material from deep within the Earth to the surface.
"The kimberlite eruption carries the diamonds from depths that we can't access up to the surface, and that's how we can study them," Halpin explained. "It's like a window into deep earth processes."
The researchers looked at tiny inclusions of other material contained within the diamonds, which offered insight into the composition of the Earth's mantle and other dynamics at a depth underground that today's scientists cannot reach.
That involved the use of specialized, high-tech equipment, such as an electron microprobe that uses a focused electron beam to ascertain the chemical composition of minerals. A high-resolution technique known as nanoscale secondary ion mass spectrometry (NanoSIMS) detected carbon isotopes within the diamonds, while cathodoluminescence spectroscopy gave insight into the diamonds' growth history.
The researchers also used Fourier Transform Infrared Spectroscopy (FTIR), which analyzes how minerals absorb infrared light to identify chemical bonds and molecular structures. Specifically, they looked at how much nitrogen the diamonds contained, which allowed them to estimate how long the diamonds spent in the mantle and the temperature they experienced.
Before the various types of scans, the diamond samples need to be prepared - hence the polishing.
"Given that diamonds are the most scratch-resistant mineral in the world, polishing was no simple task. One of the major challenges I faced this summer was learning how to prepare diamond samples for analysis," Halpin explained. "This involved a significant amount of trial and error using various techniques, and ultimately using a diamond polishing wheel and ion milling machine."
Next steps
Diamonds aren't the only mineral involved in the researchers' analysis; the Carnegie scientists also looked at garnets in the host rock, since garnets form under similar pressures and temperatures as diamonds.

They discovered something interesting: the inclusions inside the diamonds revealed chemical fingerprints pointing to subducting oceanic crust and recycled sediments, while the host rock had a different origin. This information sheds light on the complex processes that create diamonds, a question researchers are still working to fully understand.
"The diamonds formed under completely different conditions than the host rocks did and at different times," Halpin explained. "We're still trying to understand the processes of formation."
During the internship, Halpin collaborated with both peers and Carnegie scientists. Interns gave weekly presentations about their research and attended seminars on scientific communication, graduate school and how to write scientific abstracts; they also learned about research conducted by Carnegie scientists, from monitoring volcanic eruptions to measuring the Earth's magnetic field.
Back at Binghamton, Halpin is continuing to work on the Carnegie research project remotely, analyzing data, helping prepare a manuscript for publication, and creating a research poster. In December, she will share her findings in New Orleans at the 2025 American Geophysical Union's annual meeting, the largest international earth and space conference in the world.
It will be her first conference, and she's looking forward to reconnecting with her fellow interns there.
"It's been such a great summer," Halpin reflected. "I'll definitely never forget it."
She'll also use what she learned about ancient geologic processes in her senior thesis. Working with Distinguished Professor Tim Lowenstein, she is also exploring fluid inclusions in salt crystals, which provide insight into the composition of ancient seawater.
"Kate Halpin's lab work and depth of understanding stand with the very best students in Earth Science. It is great to see Kate get a career start at Carnegie Science and come back to Binghamton, a 'new' person enthusiastic to begin a senior thesis project," Lowenstein said.
This fall, she plans to apply to doctoral programs in geochemistry. She's open to whatever the future brings, whether that entails teaching, research or both, she said.
"Many researchers collaborate with NASA to answer questions in both geochemistry and planetary science. I've always been fascinated by space, and this is something that I would love to explore further," Halpin said. "At Carnegie, I met scientists who utilize meteorites to reveal clues about the formation of the solar system and the history of planets, including our own. If I could do that in some capacity, it would be incredibly fulfilling."