Autophagy is a fundamental cellular process by which cells capture and degrade their own dysfunctional or superfluous components for degradation and recycling. Recent research has revealed that phase separated droplets have a range of important functions in cells. An international collaboration between German, Norwegian, and Japanese researchers has unravelled the mechanisms underpinning both how these droplets are captured through autophagy, as well as how droplets can serve as a platform from which structures facilitating cytosolic autophagy arise.
Autophagy 'eats' portions of liquid droplets in cells
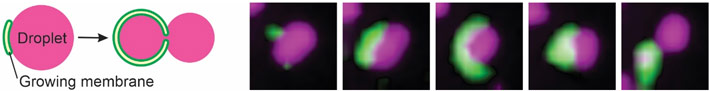
A liquid droplet made of phase-separated proteins (magenta) can associate with autophagy membranes (green). In this paper, it was shown that the droplet-membrane interaction depends on wetting and is defined by the surface tension of the droplet. As autophagy membranes expand on the droplet surface, droplets of sufficiently low surface tension are unable to overcome the intrinsic curvature of the membranes and subsequently deform. This results in a subset of the droplet being 'bitten off' by autophagy. The images at the right show this process occurring in a cultured human cell.
Two worlds meet
Autophagy[1], a critical intracellular degradation pathway that plays a key role in human health, has attracted the attention of cell biologists for decades, culminating in the award of the 2016 Nobel Prize in Physiology or Medicine to Tokyo Institute of Technology (Tokyo Tech) Specially Appointed Professor Yoshinori Ohsumi in 2016 for his work uncovering the mechanisms of this process. Recently, the autophagy has been observed to degrade fluid droplets[2], which are formed by phase separation and have been identified as important structural components of cells in rapidly progressing research. But how this 'eating' of fluid droplets occurs is unknown.
This simple but important question prompted Dr Roland Knorr at the University of Tokyo to assemble an international team of researchers from Göttingen (Germany), Oslo (Norway), and Tokyo (Japan), including Dr Alexander I. May from the Institute of Innovate Research at Tokyo Tech. This group set out to understand the biological process of autophagosomal droplet sequestration, discovering that an intricate physical mechanism underlies the relationship between autophagy and droplets. Their results, published in this week's issue of Nature
 , represent a major breakthrough in our understanding of how autophagy captures cellular material and how droplets are degraded in cells. These findings promise to inform therapeutic studies targeting autophagy and the abnormal accumulation of droplet materials observed in neurodegenerative and other diseases.
, represent a major breakthrough in our understanding of how autophagy captures cellular material and how droplets are degraded in cells. These findings promise to inform therapeutic studies targeting autophagy and the abnormal accumulation of droplet materials observed in neurodegenerative and other diseases.
One bite at a time
In the first step of autophagy, the isolation membrane, a key functional structure of autophagy made up of a double-layered lipid membrane shaped somewhat like a flattened tennis ball, grows in size, bends to form a cup-like shape and ultimately forms a spherical structure known as the autophagosome. Autophagosomes capture cytosolic and other cellular material such as droplets, isolating this cargo from the rest of the cytosol, following which the cargo is broken down and its building blocks recycled by the cell. The researchers focused on the isolation of droplets, which they found can be understood in terms of surprisingly simple and fundamental physical principles.
Droplets are spherical due to the effect of surface tension, which acts to minimize a droplet's surface area. How strongly a droplet can resist deformations from a spherical shape is defined by the droplet's surface tension[3], the value of which reflects how strongly the droplet and the surrounding cytosol repel each other. Critically, lipid membranes are able to sit at the interface between the droplet and cytosolic fluids, a phenomenon known as wetting. Wetting depends on how strong a membrane favours interaction with the droplet and the cytosol, as well as the droplet surface tension.
The researchers developed a theoretical model that accounts for these physical forces to explain how autophagy membranes interact with and capture droplets. They found that the shape of the droplet-isolation membrane pair is governed by a competition between the droplet's resistance to deform and the tendency of the isolation membrane to bend. Dr. May explains how physical forces determine the outcome of the droplet-isolation membrane interactions: "During the initial phase of autophagy, isolation membranes on droplets are small, which means they only have a weak tendency to bend. As the membrane area grows, however, these membranes become more likely to bend – the bending energy increases. The droplet's surface tension defines its resistance to deformation, and if the surface tension is low enough a critical point can be reached where the bending energy of the isolation overcomes the droplet's surface tension. In this case, a piece of the droplet is 'bitten off' and captured within an autophagosome. If this critical point is never reached and the surface tension of the droplet 'wins' this competition by overcoming the membrane bending energy, the isolation membrane will continue to grow along the droplet surface, eventually engulfing the entire droplet. Droplet autophagy can therefore be thought of as a sort of tug-of-war between the droplet's surface tension and the isolation membrane's bending energy."
With the model predicting this trade-off between 'piecemeal' and 'complete' autophagy, the team set out to confirm these findings in living cells. The researchers used a cutting-edge combination of fluorescence and electron microscopy to follow droplet compartments that enrich a protein called p62 or SQSTM1[4]. As predicted by modelling of low surface tension droplet conditions, the localisation of small isolation membranes to the droplet surface was followed by the 'biting off' of pieces of droplet. But the team needed to develop an innovative means of controlling droplet surface tension to confirm the influence of droplet properties on sequestration.
Autophagy on demand
To address this question, the researchers devised a minimal synthetic experimental system that eliminates the complexity of the intracellular environment. Using this approach, they observed the self-assembly of isolation membrane-like structures from pre-existing membranes on the surface of droplets with high surface tension. The tuneable nature of this experimental setup allowed the researchers to decrease droplet surface tension, thereby testing what effect this has on droplet capture. As predicted by the model, they observed that flattened isolation membranes transform via an intermediate cup-like shape into an autophagosome-like structure, thereby taking a bite from the droplet. Together, these results confirm the veracity of the model and demonstrate that wetting is the physical mechanism governing autophagosome formation at droplets.
These results indicate that biologists are still exploring only the tip of the iceberg when it comes to the significance of phase separation in autophagy. Intriguingly, another study
 published in Nature last year that was co-authored by Dr. Ohsumi, Dr. Knorr and Dr. May showed that the site of autophagosome formation in yeast cells is in fact a fluid droplet that is never captured. Dr. Knorr remarks: "I was very fascinated to discover droplets being a novel key autophagy structure. Now, we wanted to understand the mechanism behind our observation that some types of droplets are degraded by autophagosomes, such as p62, but others not, including the site of autophagosome formation."
published in Nature last year that was co-authored by Dr. Ohsumi, Dr. Knorr and Dr. May showed that the site of autophagosome formation in yeast cells is in fact a fluid droplet that is never captured. Dr. Knorr remarks: "I was very fascinated to discover droplets being a novel key autophagy structure. Now, we wanted to understand the mechanism behind our observation that some types of droplets are degraded by autophagosomes, such as p62, but others not, including the site of autophagosome formation."
Switching things up
The simple competition between isolation membrane bending and droplet surface tension described above assumes that the properties of the isolation membrane aren't altered when it sticks to the droplet surface. This is unlikely as each side of the isolation membrane wets two very different fluids during droplet autophagy: the droplet or the cytosol. The team expanded on their model to account for this, finding that such wetting-derived intrinsic asymmetry of isolation membranes determines bending direction and thereby the material captured for degradation: either the droplet via the piecemeal pathway, or the cytosol through the growth of the isolation membrane away from the droplet. The upshot of this is that the particular combination of isolation membranes, droplet properties and cytosolic state combine to specify the droplet as a target for autophagy or, counterintuitively, as a platform that enables autophagy of the surrounding cytosol.
To test this, the researchers modified the p62 protein to lack a specific motif that is known to interact with the proteins in the isolation membrane, thereby weakening the isolation membrane-droplet association. This manipulation had a radical effect: while isolation membranes were initially observed to grow along p62 droplets in wild-type (unmodified) cells, they instead bent to capture cytosol, leaving the droplet completely intact. Tiny changes in droplet properties therefore have critical implications for the mode of autophagy in living cells, specifying piecemeal or complete enclosure of droplets, and even the capture of cytosolic material.
Elucidation of the underlying physical rationale that enables this switch provides an entirely new perspective in our understanding of the mechanism of autophagy, as well as the role of droplets and physical principles such as wetting in cells. This understanding lays the groundwork for a host of new studies on the implications of physical forces in cell biology, as well as providing new clues that will help understand how autophagy is involved in diseases that are not easily treated, such as neurodegenerative diseases and cancer.






