Scientists at Tokyo Institute of Technology (Tokyo Tech) developed a hybrid material constructed from a metal oxide nanosheet and a light-absorbing molecule for splitting water molecules (H2O) to obtain dihydrogen (H2) under sunlight. Since H2 can be used as carbon-free fuel, this study provides relevant insight towards clean energy generation.
In line with the depletion of fossil fuels and the environmental problems our planet faces due to their combustion, developing technology for clean energy generation is a topic of global interest. Among the various methods proposed to generate clean energy, photocatalytic water splitting is showing much promise. This method utilizes solar energy to split water (H2O) molecules and obtain dihydrogen (H2). The H2 can then be used as a carbon-free fuel or as raw material in the production of many important chemicals.
Now, a research team led by Kazuhiko Maeda at Tokyo Tech has developed a new photocatalyst consisting of nanoscale metal oxide sheets and a ruthenium dye molecule, which works according to a mechanism similar to dye-sensitized solar cells. While metal oxides that are photocatalytically active for overall water splitting into H2 and O2 have wide band gaps, dye-sensitized oxides can utilize visible light, the main component of sunlight (Figure 1). The new photocatalyst is capable of generating H2 from water with a turnover frequency of 1,960 per hour and an external quantum yield of 2.4 %.
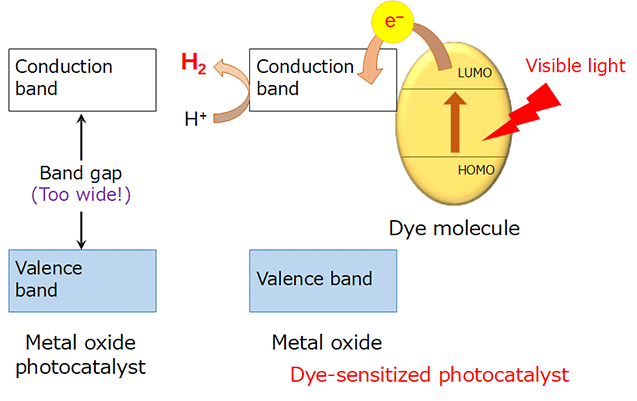
Figure 1. Dye-sensitized H2 evolution using a wide-gap metal oxide.
These results are the highest recorded for dye-sensitized photocatalysts under visible light, bringing Maeda's team a step closer to the goal of artificial photosynthesis ― replicating the natural process of using water and sunlight to sustainably produce energy.
The new material, reported in Journal of the American Chemical Society, is constructed from high-surface-area calcium niobate nanosheets (HCa2Nb3O10) intercalated with platinum (Pt) nanoclusters as H2-evolving sites. However, the platinum-modified nanosheets do not work alone, as they do not absorb sunlight efficiently. So a visible light-absorbing ruthenium dye molecule is combined with the nanosheet, enabling solar-driven H2 evolution (Figure 2).
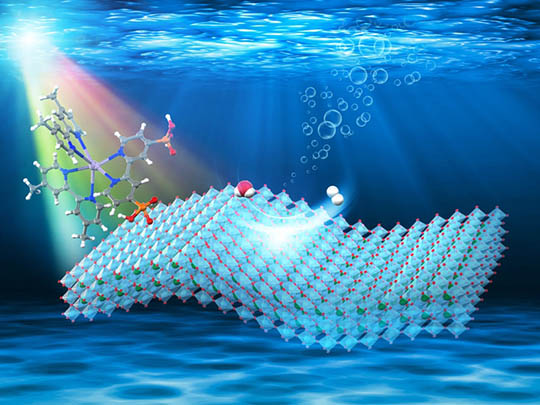
- Figure. 2
- An illustration of visible-light-driven H2 evolution on ruthenium-dye/HCa2Nb3O10 nanosheet. Adopted with permission. Copyright 2020, American Chemical Society.
What makes the material efficient is the use of nanosheets, which can be obtained by chemical exfoliation of lamellar HCa2Nb3O10. The high-surface-area and structural flexibility of the nanosheets maximize dye-loadings and density of H2 evolution sites, which in turn improve H2 evolution efficiency. Also, to optimise performance, Maeda's team modified the nanosheets with amorphous alumina, which plays an important role in improving electron transfer efficiency. "Unprecedentedly, the alumina modification for nanosheets promotes dye-regeneration during the reaction, without hindering electron injection from the excited-state dye to the nanosheet ― the primary step of dye-sensitized H2 evolution," Maeda says.
"Until just recently, it was considered very difficult to achieve H2 evolution via overall water splitting under visible light using a dye-sensitized photocatalyst with high efficiency," explains Maeda. "Our new result clearly demonstrates that this is indeed possible, using a carefully designed molecule-nanomaterial hybrid."
More research still needs to be done, as it will be necessary to further optimize the design of the hybrid photocatalyst to improve the efficiency and long-term durability. Photocatalytic water splitting may be a crucial means of meeting society's energy demands without further harming the environment, and studies like this one are essential stepping stones to reaching our goal of a greener future.






