Boron agents termed GluBs, developed by Science Tokyo researchers, overcome a key limitation in cancer therapy by entering tumor cells through a pathway that standard drugs cannot use. The GluBs target ASCT2, a transporter abundant in aggressive cancers such as glioblastoma and breast cancer, rather than the LAT1 route. Results from cell and animal studies show the agents were safe and effective in limiting tumor growth, indicating potential to treat cancers with limited LAT1 expression.
Expanding Boron Neutron Capture Therapy with ASCT2-Guided Boron Delivery
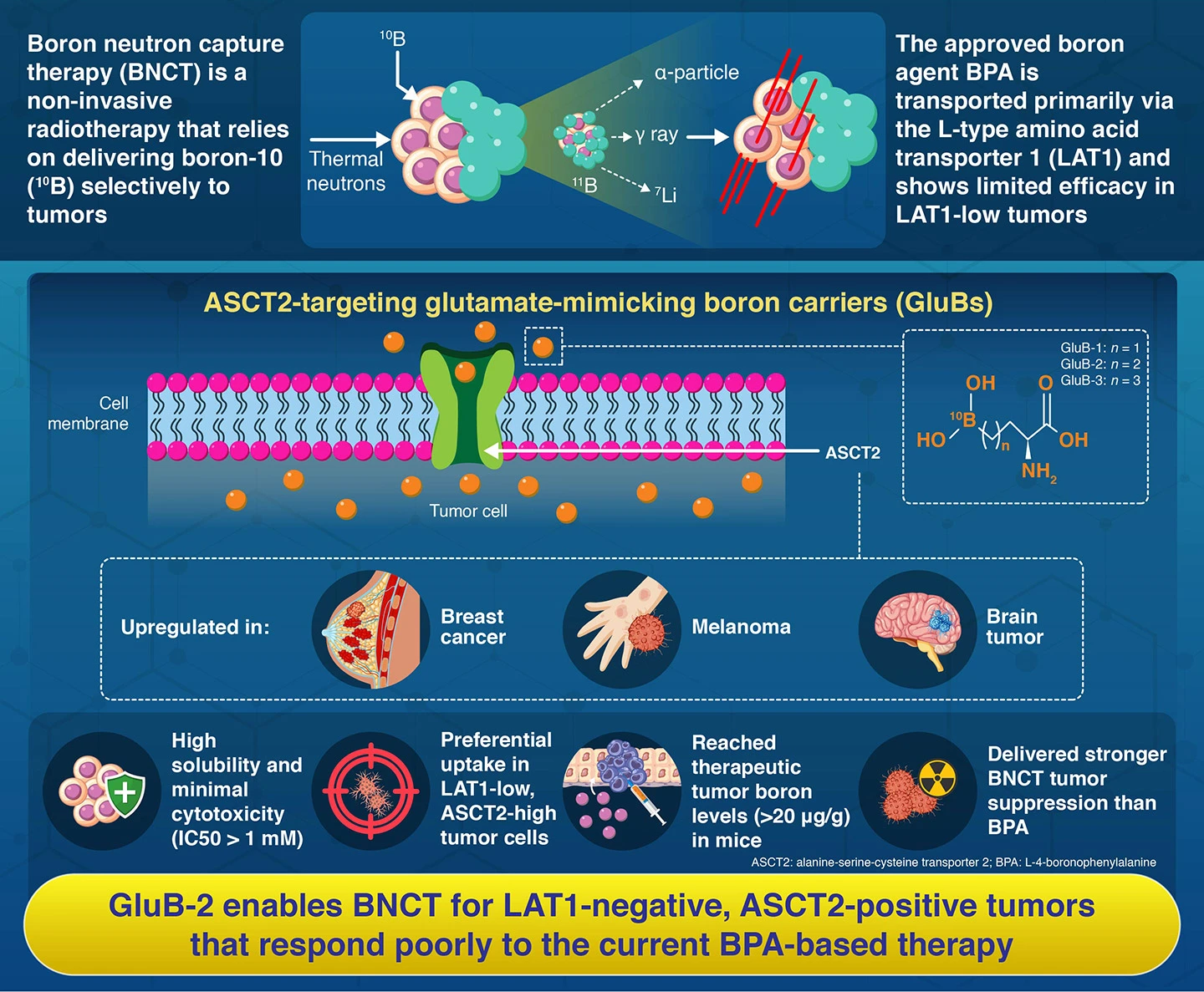
Boron neutron capture therapy (BNCT) is a non-invasive form of radiotherapy that works by delivering enough boron-10 directly to tumor cells. When these boron-loaded cells are exposed to low-energy neutrons, they release highly localized bursts of energy that destroy the cancer cells while largely sparing nearby healthy tissue.
In current clinical practice, the most widely used boron carrier is L-4-boronophenylalanine (BPA). BPA enters tumors primarily through the L-type amino acid transporter 1 (LAT1), a protein that is overexpressed in many cancers. However, tumors with low LAT1 expression respond poorly, leaving a significant group of patients without effective treatment options and creating an urgent need for boron agents that utilize alternative uptake pathways.
To address this limitation, researchers from Institute of Science Tokyo (Science Tokyo), Japan, developed glutamate-mimicking boron carriers called GluBs. These compounds are designed to enter cancer cells through the alanine-serine-cysteine transporter 2 (ASCT2), a nutrient transporter that supports the metabolic demands of rapidly proliferating tumors. ASCT2 is abundant in several difficult-to-treat cancers, including triple-negative breast cancer, melanoma, and glioblastoma, making it an attractive new gateway for BNCT drugs.
The breakthrough was achieved by a research team led by Professor Hiroyuki Nakamura and Assistant Professor Kazuki Miura from the Laboratory for Chemistry and Life Science, Science Tokyo, in collaboration with Professor Minoru Suzuki from the Institute for Integrated Radiation and Nuclear Science, Kyoto University, Japan. The findings of this study were made available online on December 22, 2025, and were published in Volume 390 of the Journal of Controlled Release on February 10, 2026.
"These small-molecule boron agents, or the GluB series, demonstrated efficacy against tumors poorly responsive to the clinically used BNCT agent BPA, addressing a significant limitation of current boron neutron capture therapy," says Nakamura.
The team synthesized three related molecules, GluB-1, GluB-2, and GluB-3, each with progressively longer linkers between the amino acid backbone and the boron atom. In laboratory tests, all three dissolved readily in water and showed low toxicity at doses relevant for BNCT, a crucial requirement because such agents must be administered in relatively large quantities during treatment.
When the scientists measured uptake across multiple tumor cell lines, the GluBs preferentially accumulated in cancers with high ASCT2 and low LAT1 expression. The advantage was particularly evident in human breast cancer and glioblastoma cells, which are often resistant to BPA. Among the candidates, GluB-2 demonstrated the most favorable balance of safety and tumor targeting.
The researchers next examined performance in mice bearing CT26 colon tumors and human U87MG glioblastoma xenografts, both models known for limited responsiveness to BPA. GluB-2 delivered boron to tumors at concentrations of 21 µg[B]/g within hours of administration, meeting the threshold dosage for BNCT (20 µg[B]/g).
On neutron irradiation, tumors treated with GluB-2 showed markedly stronger growth suppression than those given BPA. In the U87MG model, tumor-to-normal and tumor-to-blood ratios, which indicate how selectively boron concentrates in cancer tissue, reached or exceeded 3.0, values considered encouraging for predicting clinical usefulness in humans. Throughout the tests, the mice maintained their weight and showed no obvious signs of damage to major organs, suggesting the treatment was well tolerated at the doses examined.
"Our study demonstrates that GluB-2 enables therapeutic small-molecule delivery of boron-10 to tumors through multiple dosing routes and may expand the clinical applicability of BNCT beyond BPA," Nakamura concludes.
This work represents the first successful attempt to design BNCT boron carriers around the ASCT2 transporter. By opening a new pathway into cancer cells, the method offers a potential lifeline for patients whose tumors do not respond to the existing therapy.
Reference
- Authors:
- Kazuki Miura1,2, Tomoyuki Araki3, Taiki Morita3,4, Kai Nishimura2, Satoshi Okada1,2, Minoru Suzuki5, Hiroyuki Nakamura1,2
- Title:
- Alanine-serine-cysteine transporter-targeted small-molecule boron carriers for neutron capture therapy of L-4‑boronophenylalanine-refractory tumors
- Journal:
- Journal of Controlled Release
- Affiliations:
- 1Laboratory for Chemistry and Life Science, Institute of Science Tokyo, Japan
2School of Life Science and Technology, Institute of Science Tokyo, Japan
3School of Life Science and Technology, Institute of Science Tokyo, Japan
4Laboratory for Chemistry and Life Science, Institute of Science Tokyo, Japan
5Institute for Integrated Radiation and Nuclear Science, Kyoto University, Japan






