Walking is the most natural of movements. Without thinking, we put one foot forward and then the next, on and on, propelling us forward. So, if we're not consciously directing this complex interplay of nerves and muscles, what is?
"As one might expect, it's the brain that initiates locomotion. But it doesn't coordinate it," says Columbia neuroscientist George Mentis, PhD, who investigates the circuits that control walking with an eye toward finding new treatments for patients with ALS, SMA, and spinal cord injuries.
Coordination of our many walking muscles is handled by neurons in the spinal cord, says Mentis, associate professor of pathology & cell biology (in neurology) at the Columbia University Vagelos College of Physicians and Surgeons.
It's a complex job: With precise timing, these neurons must send signals so the left and right leg alternate their activity-left, right, left, right-and so flexor and extensor muscles in each leg contract in an alternating fashion.
Most scientists thought that such a complex task could only be handled by complex neuronal circuits, with contributions from different types of neurons. This assembly of circuits, called the central pattern generator, seemed to run the show.
But Mentis's newest research reveals that just a single type of neuron within this assembly of circuits is completely responsible for keeping our legs in lockstep.
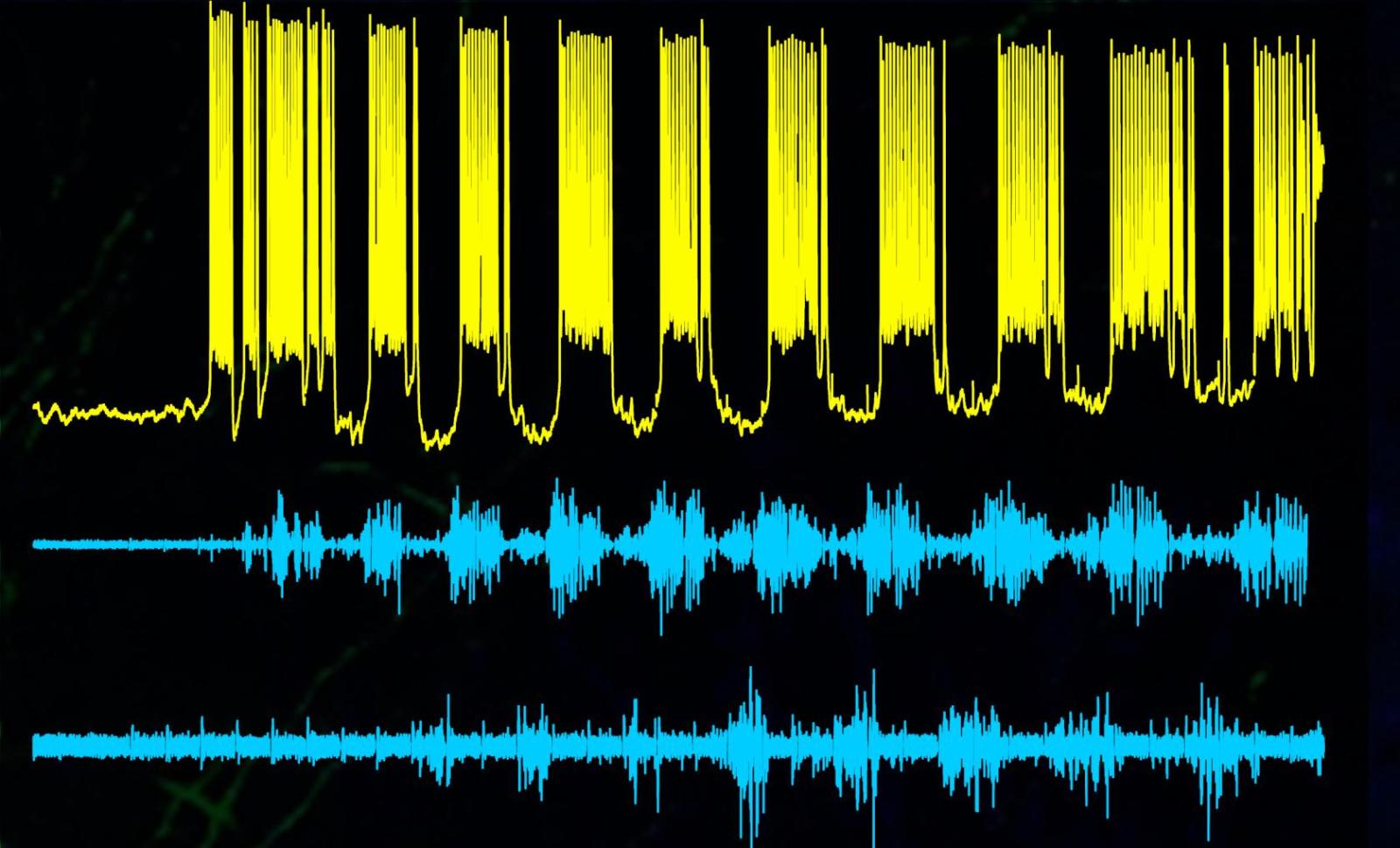
And like tiny drill sergeants, without these neurons collectively commanding, "left, right, left, right," we'd never get anywhere.
The neurons-properly known as ventral spinocerebellar tract neurons-make contacts with other spinal cord neurons and orchestrate the ability to move muscles.
In the new study, Mentis and his colleagues found that when just these cells were chemically silenced in freely moving adult mice, the animals could no longer move properly. After the drugs wore off, normal movement returned. Additionally, activation of these cells by either light or drugs can induce locomotor behavior in juvenile mice. "In other words, these neurons are both necessary and sufficient for locomotor behavior," says Mentis, whose findings were published Jan. 20 in Cell.
"It may not be enough to reconnect the brain and the spinal cord in people with severed spinal cords. Everything has to be tightly balanced between exciting certain neurons and inhibiting others. If this balance is compromised, you won't have coordinated movement."
Mentis also found that the cells are highly interconnected, a property that likely contributes to their ability to generate the complex rhythmic patterns necessary for locomotion.
The findings have important implications for the development of new therapies for people with spinal cord injuries or motor disorders.
"For example, it may not be enough to reconnect the brain and the spinal cord in people with severed spinal cords," Mentis says. "Our findings suggest that you would also have to restore proper activity in the ventral spinocerebellar tract neurons to ensure that the central pattern generator is working properly. Everything has to be tightly balanced between exciting certain neurons and inhibiting others. If this balance is compromised, you won't have coordinated movement."
References
More information
The paper is titled "Control of mammalian locomotion by ventral spinocerebellar tract neurons."
All authors: Joshua I. Chalif (Columbia), María de Lourdes Martínez-Silva (Columbia), John G. Pagiazitis (Columbia), Andrew J. Murray (University College London), and George Z. Mentis (Columbia).
The research was supported by the NIH (grants R01-NS078375, R21-NS079981, R01-AA027079, and F30NS098551), the SMA Foundation, and Project-ALS.






