Scientists have made a major breakthrough in understanding how the parasite that causes malaria is transmitted from mosquitos, which could be a vital clue in discovering how it has evolved, and how it can be stopped.
In the new study, published in the journal eLife, scientists have uncovered the role of a unique cyclin-cyclin dependent protein complex that is required for rapid growth of the male cell of the malaria parasite within gut of the female mosquito. This is crucial for parasite transmission.
The research is co- led by Rita Tewari, Professor of Parasite Cell Biology in the School of Life Sciences at the University of Nottingham and Professor Mathieu Brochet at the University of Geneva.
Malaria is one of the world's biggest killers and is responsible for almost half a million deaths a year, mainly in tropical developing countries. The disease is caused by the parasite Plasmodium and is spread through the bite of an infected mosquito.
All organisms reproduce and multiply to propagate and survive. This multiplication is controlled by some specific proteins like cyclins and their partner kinases to occur at the right time during their proliferation.
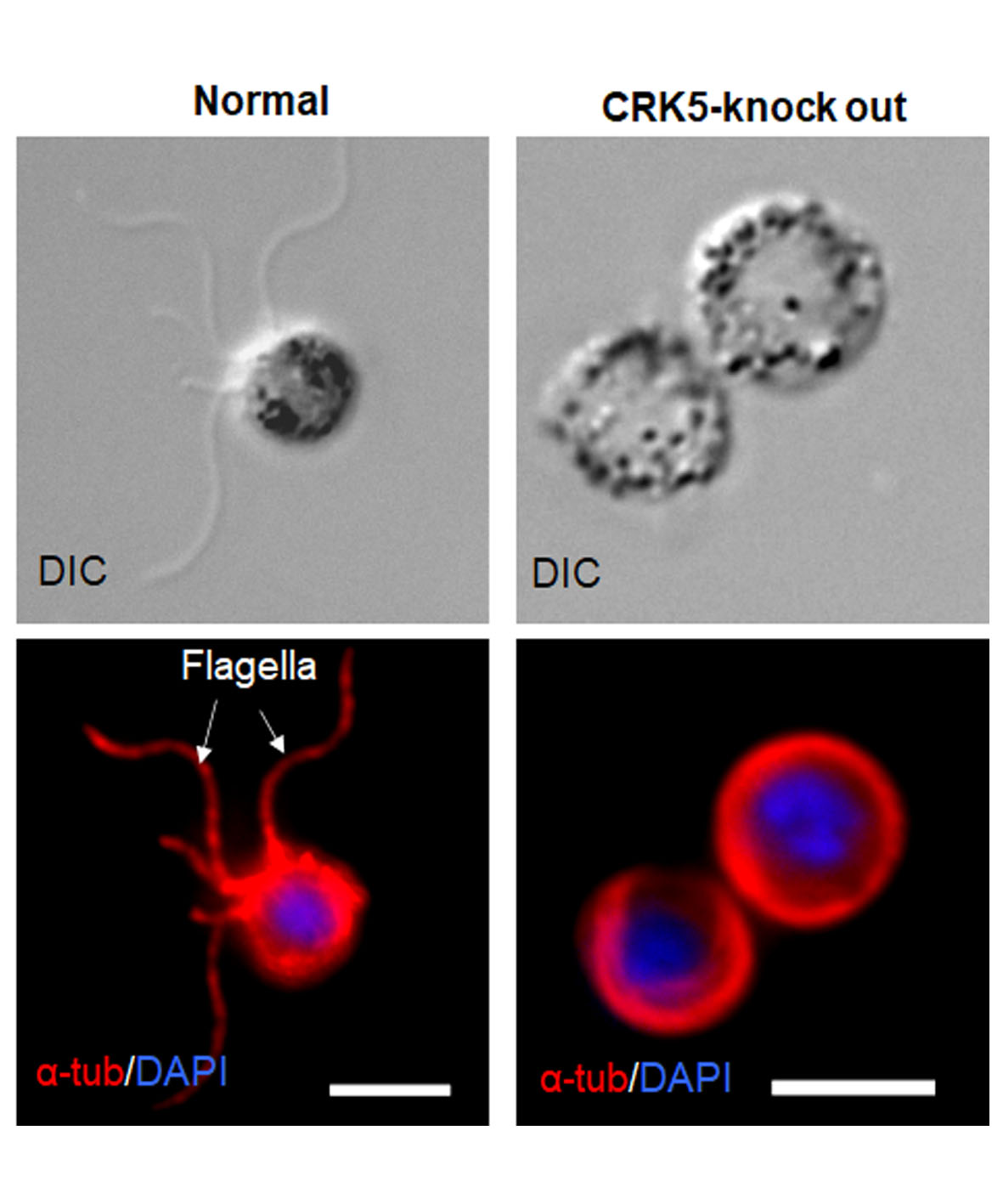
In this latest study, scientists have identified malaria specific cyclin and cyclin dependent kinases complex for male cell multiplication. The male cell in malaria is picked up by a mosquito when it feeds on the infected humans. This cell, which is called male gametocyte, undergoes accelerated growth and divides within 15 minutes to give rise to eight offspring that are flagellated sperm cells of the malaria parasite. This process is known as male gametogony. The flagellated sperm (male gamete) fertilise the ovum (female gamete) to form zygote.
This stage is crucial for parasite transmission because the flagella male cells are only produced in mosquitos.
Professor Tewari said: "We are trying to understand how the male cells grow and divide at such accelerated speeds, and the key molecules that regulate this process of multiplication. This will help us not only to understand this rapid process of parasite male sperm formation but also to identify the targets that can block the malaria transmission between humans and mosquitoes."
Prof Mathieu Brochet who co led the study added "It shows that despite some similarities with human cells, the parasites rely on significantly different strategies to proliferate"
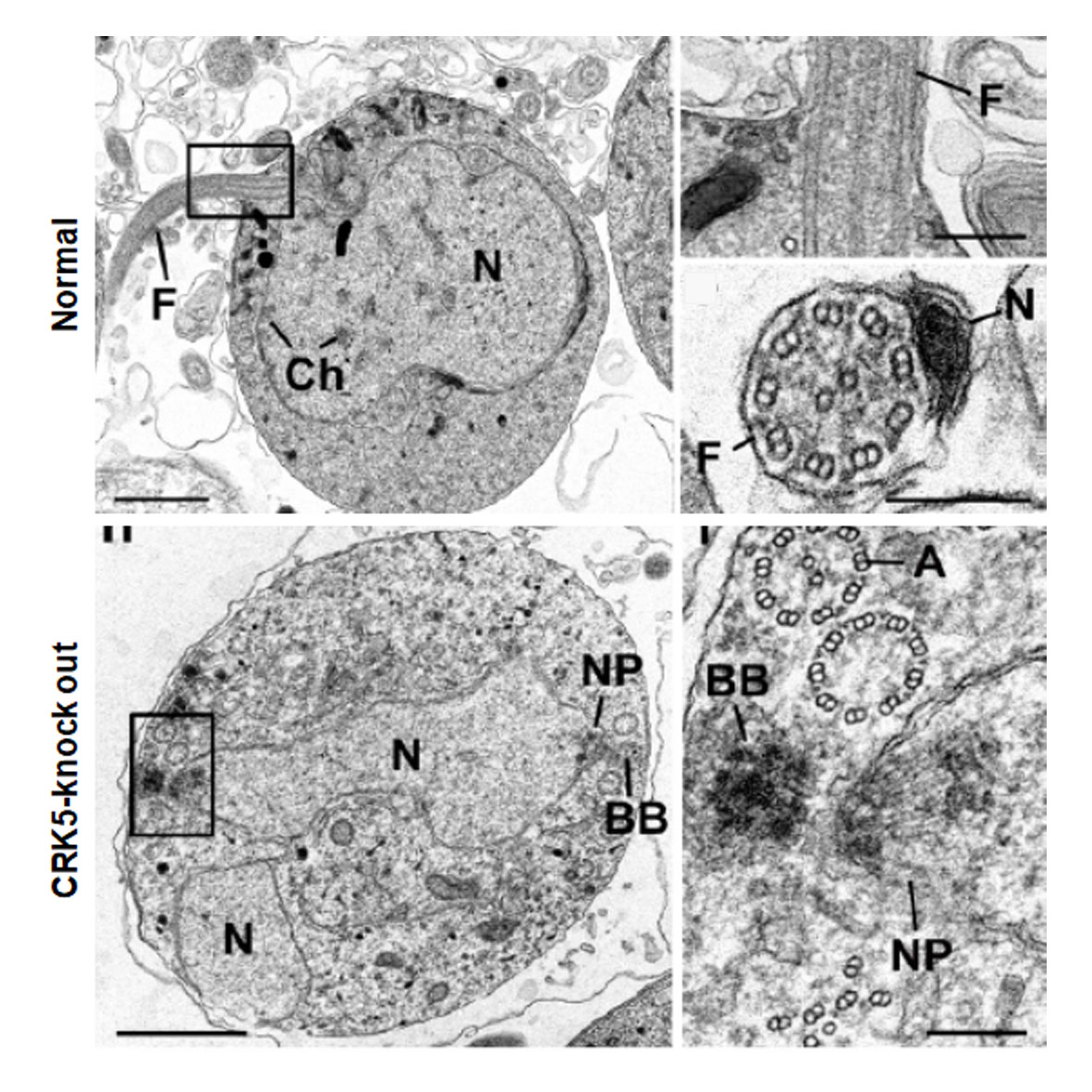
Dr M. Zeeshan, one of the lead authors of the study, said: "We have identified parasite specific complex of protein that control the male sperm multiplication. This process was severely affected when we took away the CRK5 gene which is one of the major molecules of this cyclin/cyclin dependent kinase complex. In-depth analysis shows that DNA replication and cell division was affected in male cells during sperm formation in this parasite." This could help to identify target for blocking malaria transmission by mosquito.
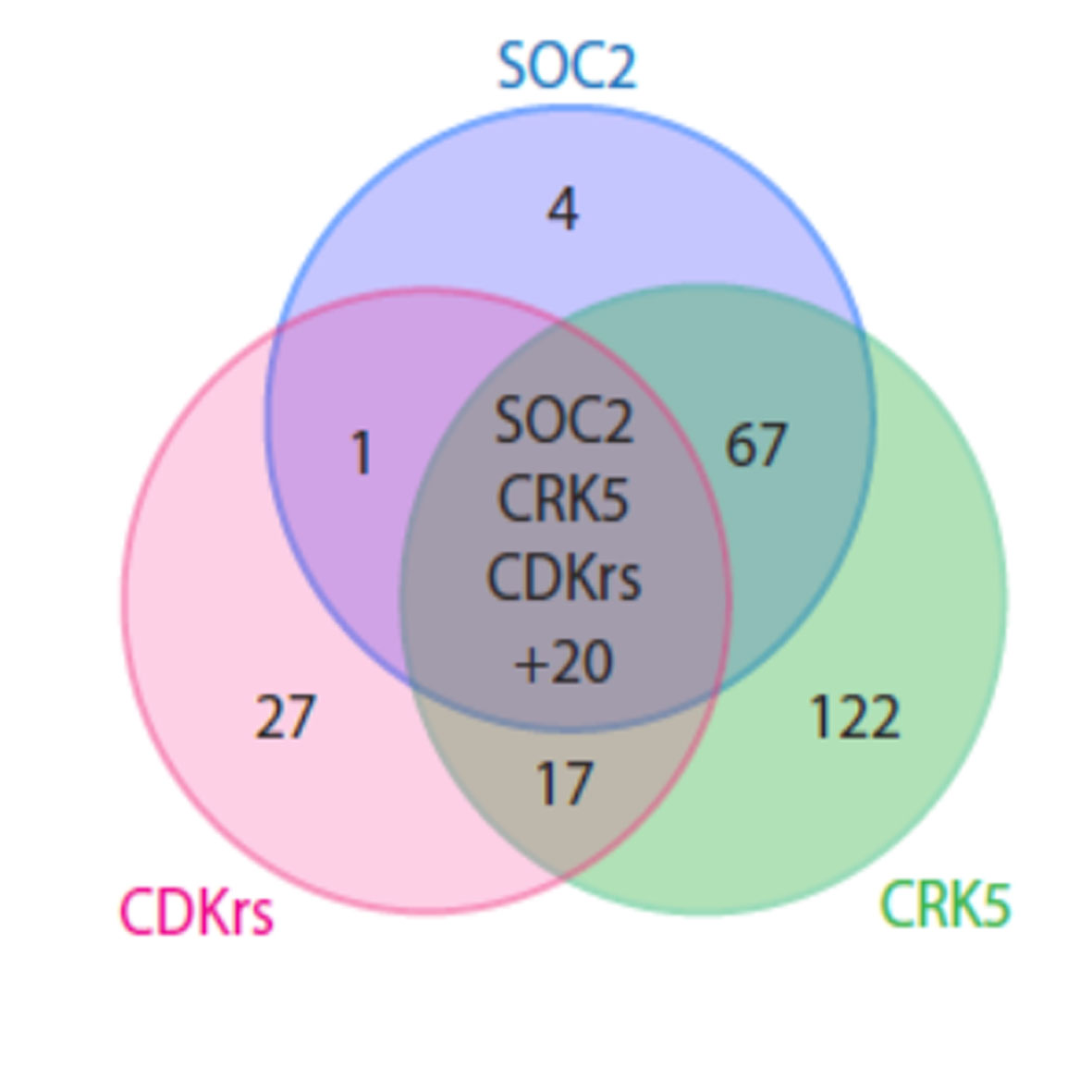
The University of Nottingham team, along with other international collaborators, also identified one molecule of this complex as a novel cyclin which is like the brake of cycle that is only present in malaria related parasites. This cyclin has unusual features and is present at different phases of the cell cycle.
Professor Tewari adds: "Most malaria research is focused on identifying therapeutic targets that can block the transmission of the disease. This study is important as it gives us a better understanding of replication and multiplication of male cells of malaria parasite. The molecules of the cyclin/cyclin dependent complex identified in this study play a crucial role in male flagella sperm formation and can be exploited as drug targets."
The research work was a collaborative effort with scientists at University of Geneva, Switzerland, Oxford Brookes University, UK; and colleagues from KAUST, Saudi Arabia and The Francis Crick Institute, London. The work was funded by BBSRC, MRC, SNF, INSERM, EMBO, CRUK and CRG-KAUST.
The full study can be found here.






