The mechanics of the onset of cancer or neurodegenerative disorders like Alzheimer's disease or ALS remain a mystery. Scientists associate these diseases with an increase in unstable molecules called reactive oxygen species (ROS) in cells, but they don't fully know why they form or why these molecules might pose a problem. They also are beginning to determine which parts of cells are to blame for producing ROS.

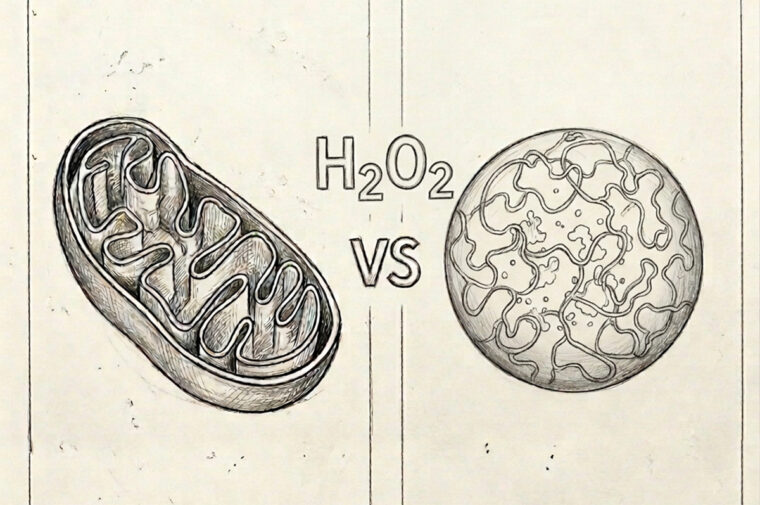
Studies have shown that one possible source of these free oxygen radicals is mitochondria, membrane-bound structures in the cell. And previous work by Yifan Dai, an assistant professor of biomedical engineering in the McKelvey School of Engineering at Washington University in St. Louis, found yet another origin for ROS: small agglomerations of mostly proteins called biomolecular condensates.
In a new paper published in the Journal of the American Chemical Society (JACS), Dai and Anthony A. Hyman, of the Max Planck Institute of Molecular Cell Biology and Genetics, dive deeper. They address how these biomolecular condensates create oxygen-based radicals without the presence of enzymes and uncover the chemical origins of the radical sources. Such findings provide the molecular basis for the study of condensates' roles in cellular physiology and diseases.
The electrochemistry in condensates
Mitochondria, long considered the primary source of free oxygen radicals, are bound by membranes and need enzymes to catalyze the formation of ROS.
In 2023, Dai and his team found that biomolecular condensates don't need to play by the same rules. They are not bound by membranes, nor do they need enzymes to kickstart redox reactions. Instead, free oxygen radical-forming reactions in condensates are spontaneous.
What drives such spontaneity? The exact sources and mechanisms for the processes were a mystery. The answer lies in the nature of the condensate itself. Unequal distribution of charged ions within the cluster creates a discrete interfacial electric field between the condensate and the rest of the cell. Such marked differences in charges from positive and negative ions result in electrical fields, which then trigger oxidation reactions.
The path to rogue behavior from the ROS or free oxygen radicals is long yet systematically facilitated by biomolecular condensates. Multiple pathways can generate hydrogen peroxide, which can contribute to protein damage and accelerate the pathway to neurodegenerative diseases.
Another important finding from Dai and his team is that the condensate environment is not static, but ages with time.
In another recent paper in JACS, Dai and Richard N. Zare, of Stanford University, explained how biomolecular condensates, with an ability to trigger spontaneous oxidation processes, can help the nitrogen cycle through its different chemical forms.
Exploring cell behavior from the lens of physical and electrochemistry is important, Dai said, because it can complement knowledge of cell genomic sequencing to better understand the basic triggers of cancer and many other diseases.






