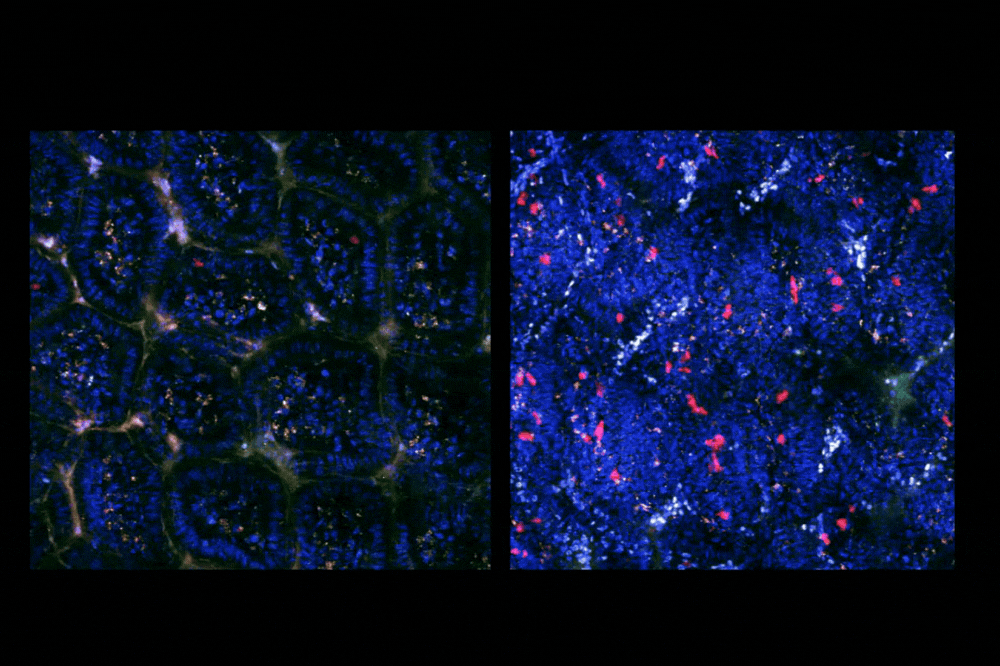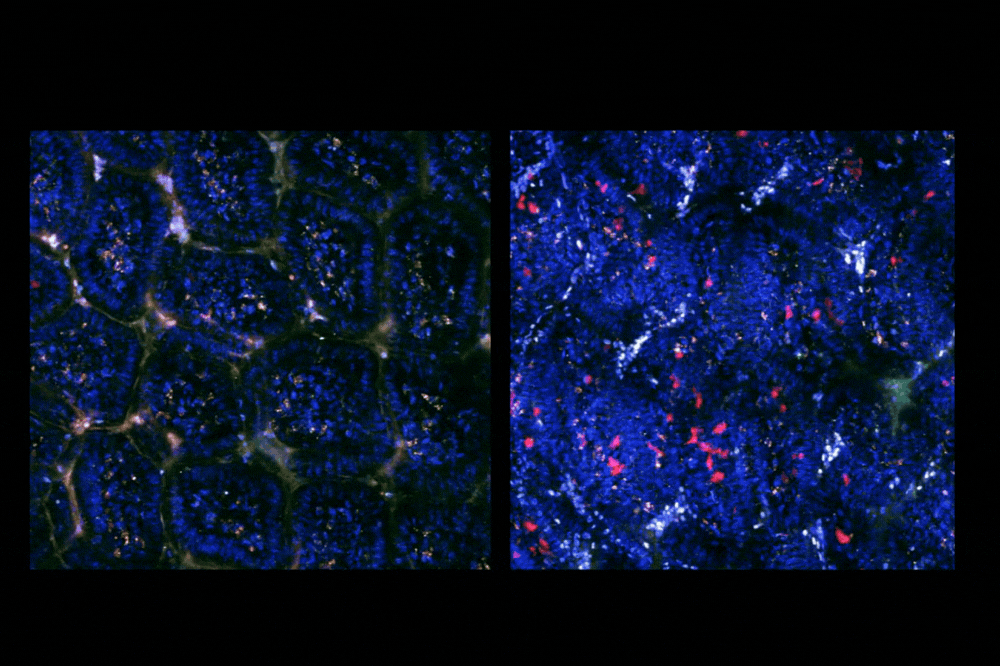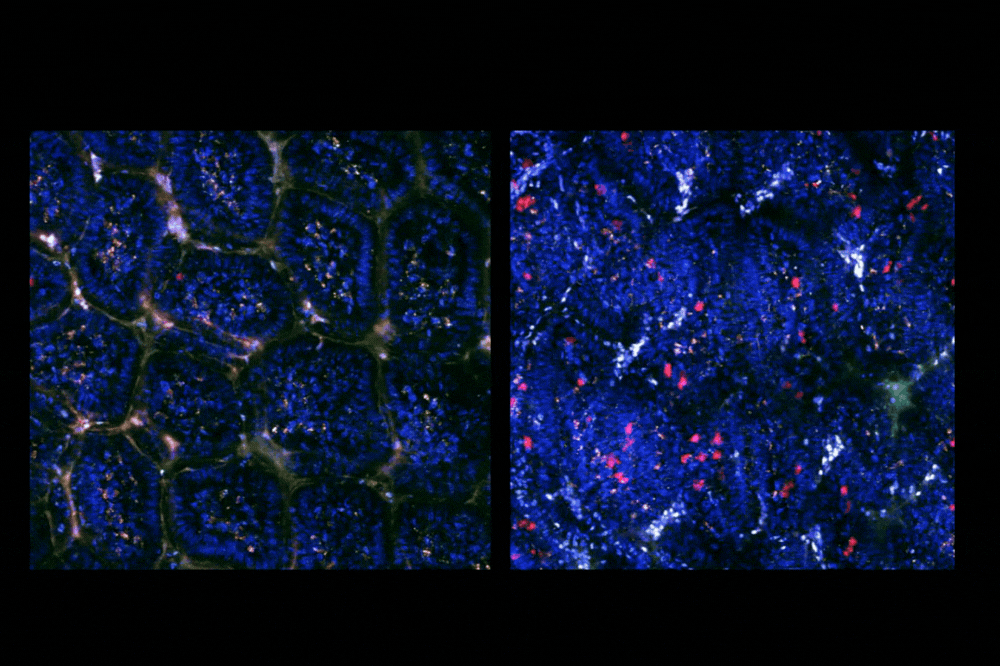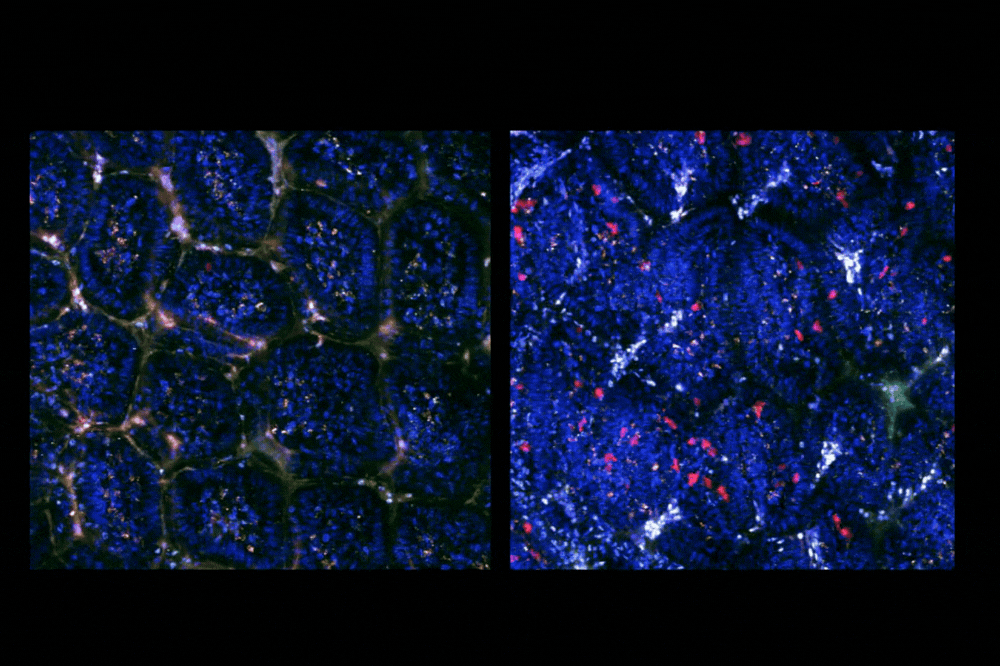
Live imaging (intravital imaging) within the intestine of TRACK mice. Left: Healthy, Right: Infected. Blue: Epithelial cells, Red: Labeled T cells responding to an enteric viral infection. (Credit: Mucida lab)
Our immune system relies on T cells to fight infections. But T cells don't just show up and react-first, they train, get a game plan, and coordinate their defenses in lymphoid organs.
Researchers have struggled to understand how this counteroffensive evolves across these sites. Now, a new tool from researchers at The Rockefeller University and Biohub allows scientists to permanently tag recently activated T cells with a fluorescent protein to track how they travel and change during an infection. The system, recently described in Nature Immunology, makes it possible to precisely characterize the T cells that respond to a specific threat and understand how their location shapes them.
The researchers used the tool, called TRACK (Tracking Recently Activated Cell Kinetics), to study T cells in the lungs, lymph nodes, and spleen as they mounted and later maintained a defense against the flu virus. Their data revealed a division of labor among these locations.
"The T cell response is compartmentalized in a very elegant way," says first author Roham Parsa, group leader of Immune Cell Dynamics and Function at Biohub in New York and adjunct assistant professor in Rockefeller's Laboratory of Mucosal Immunology, headed by Daniel Mucida. "By mapping how these programs change from tissue to tissue, we can begin to design strategies to precisely redirect immune function."
The call to action
When the adaptive immune system detects something foreign, such as viral particles, it formulates a precision attack. At the front line are T cells, which become activated after antigen-presenting cells present them with their target.
Newly mobilized T cells multiply, forming genetically identical lineages called clones that either directly attack the pathogen or coordinate with other immune cells to do so. After the danger has passed, a portion of these cells remain, serving as long-lived memory T cells that protect against future infections.
Previous studies suggested that T cell responses differ between infected tissue and lymphoid tissue, but it has been difficult to capture these variations.
"Because there are so many T cells already present, you can't home in on only the particular cells you're interested in," says Mucida, who is also an affiliated investigator at the CZ Biohub NY.
Tracking the right T cells
Parsa addressed this challenge by focusing on a fleeting combination of proteins expressed together briefly after a T cell encounters an antigen. He built the TRACK system so that when a T cell in a mouse expresses these two molecules at the same time, a glowing red label switches on. This signal allowed researchers to pinpoint the T cells responding to that antigen and follow them over time, tracking their movement between organs and how their roles evolved.
In their experiments, the researchers labeled newly activated CD4+ T cells, which coordinate other immune cells into action. They then sequenced their RNA and the genetic code of their antigen receptors. They examined these cells nine days after infection, when the immune response was at its peak, and again 56 days after infection.
Shaping identity
Differences among the three organ sites revealed an evolving, and likely strategic, response. The researchers found that the tissue where T cells first encounter the virus shapes their identity: Lung T cells become specialized frontline defenders, lymph node T cells focus on helping B cells make antibodies, and spleen T cells act as mobile reserves that spread protection across the body. Over time, these populations redistribute and converge into a shared immune memory, equipping the body for faster responses in the future.
The data also revealed that genetically identical T cell clones fulfilled different roles-assisting antibody production, killing infected cells, or providing long-term protection.
"Even though they are responding to the same pathogen, they perform different functions that are involved in different aspects of adaptive immunity," Mucida says.
However, T cells reacting to different viral proteins appeared in multiple locations, suggesting tissues do not strictly segregate responses. Instead, each site favors the expansion of certain clones, likely because distinct tissues display different viral fragments that selectively stimulate specific T cells. Over time, these differences became less pronounced as clones migrated, particularly from the lungs to the lymph nodes, leading to a more even distribution.
"I think it's a way of preparing T cells in case the pathogen is reintroduced in a different location," says Parsa.
Reprogramming the immune response
Although this study focused on flu infection, the TRACK system could be used to study cancer, vaccine responses, and autoimmune conditions, says Mucida, who has begun researching T cell responses in early stage colorectal cancer and how tumors evade them. Parsa is examining autoimmune and neurodegenerative diseases such as multiple sclerosis, in which misguided T cells damage neurons.
The researchers expect immune dynamics to differ across various conditions, and envision the TRACK tool will enable scientists to reveal patterns across many different pathogens.






