A study led by Northwestern University researchers has reported a way to observe hydrogel nano and microstructure while the hydrogel remains fully solvated. The approach reveals that methylcellulose, one of the most widely used hydrogel-forming materials in biomedical research and consumer products, organises itself into a structural architecture that standard characterisation techniques have not been able to visualize.
The material is orders of magnitude more rigid than believed and theoretical models can be used together with this new information to better predict its mechanical behaviour.
The study was published in Nature Materials on 11 March 2026.
Using the technique, variable temperature liquid cell transmission electron microscopy (VT-LCTEM), along with advanced techniques such as Small and Ultra small angle neutron scattering (SANS and USANS) at the Australian Centre for Neutron Scattering at ANSTO, the team imaged a methylcellulose hydrogel in its fully solvated state, observing its assembly in real time with minimal preparative disruption.
What they found upended the structural model that has guided the field since a landmark 2013 study. The individual fibrils that form the gel do not remain a loosely tangled network of flexible strands, like a bowl of boiled spaghetti. Instead, they bundle into much larger, substantially stiffer structures, and it is those bundles, not the fibrils, that dictate how strong the gel is.
Multiple methods confirmed that individual fibrils form as expected upon heating, then continue to assemble into bundles measuring 1 to 4 micrometres in diameter. Those bundles are roughly 100 times stiffer than the fibrils they are made of. The bundles have a persistence length, a direct measure of structural rigidity, up to a thousand times greater than what previous studies had reported for the individual fibrils
The same hierarchical structure was further confirmed by combining SANS and USANS techniques on the Quokka and Kookaburra instruments respectively, which gave multilength scale information spanning from 1 nm to 20 µm length scale.
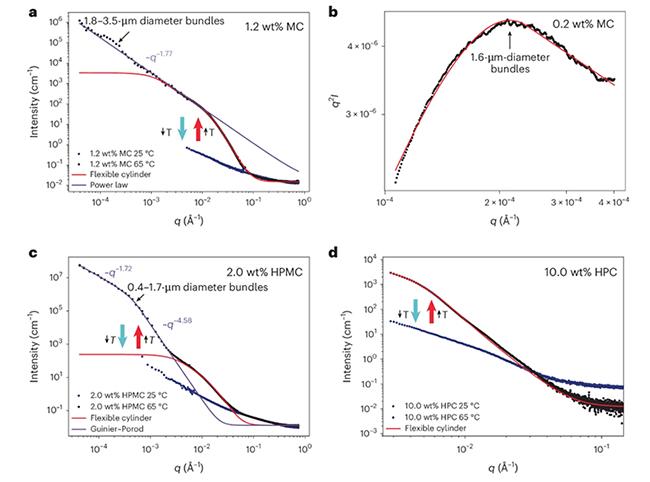
"Running these samples on our instruments across a range of temperatures and observing signals emerging at the micron length scale at higher temperatures was extremely reassuring. It provided clear evidence for a phenomenon that had previously only been predicted, as earlier SANS and SAXS measurements did not cover the larger q/size range required to detect it. Seeing this behaviour directly in the data was truly exciting," said A/Prof. Jitendra Mata, Principal Instrument Scientist, a co-author on the paper with Dr Katy Wood.
Prof Nathan C. Gianneschi, the Jacob & Rosaline Cohn Professor of Chemistry, Materials Science & Engineering, Biomedical Engineering and Pharmacology at Northwestern, who led the study, said, "We had hypothesized that if we imaged hydrogels in their liquid state with high resolution TEM, we would learn new things about their structures, not possible when they are dried or diluted for conventional imaging. It was one thing to speculate about it and another to really see these materials form under those conditions. The initial images really surprised us and excited us to dig deeper into what could be a generalizable approach to elucidating structure and function for hydrogel materials."
Northwestern University PhD candidate Nathan Rosenmann was first author on the publication.
However, the authors noted that further work is needed to fully optimise it. The long-standing gap between structure and function for these materials was not a flaw in the model. It was a consequence of underestimating the extent of the hierarchical structures involved.
Other collaborators included Case Western Reserve University, University of Florida, and Argonne National Lab.
This summary was extracted from media information provided by Northwestern University.






