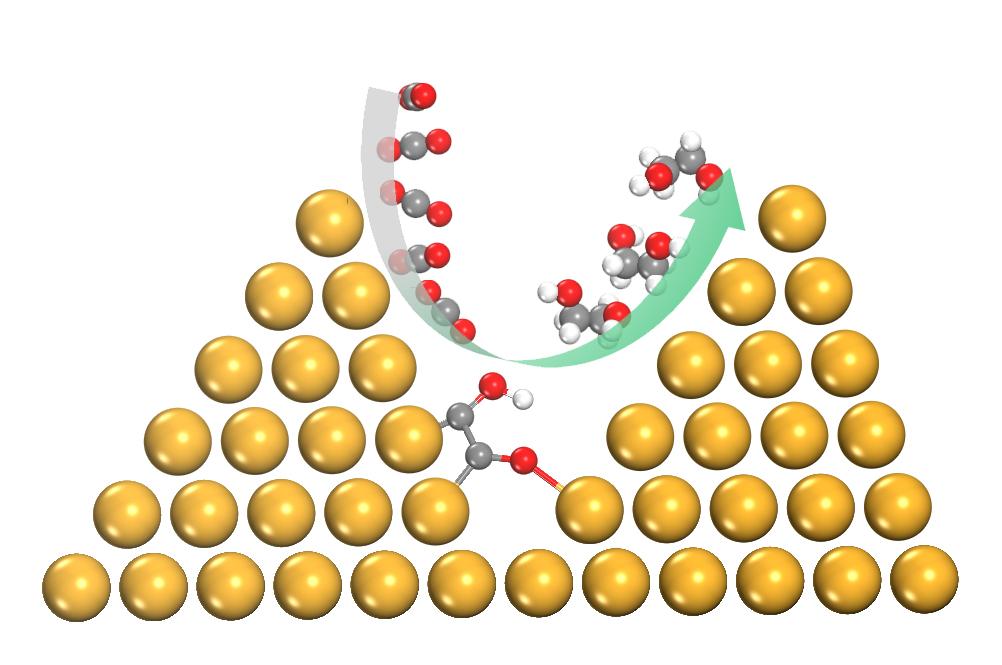
The unique spatial-confinement induced by copper nanopyramids is crucial to selectively generating ethylene glycol through a new reaction pathway. Image: Shizhang Qiao, The University of Adelaide.
Catalysts could prove to be the key to converting carbon dioxide and carbon monoxide into value-added products but their effectiveness depends on them being selective in how they work.
Scientists at the University of Adelaide are inventing better materials to make the next generation of catalysts that will assist in creating alternative fuels that could help to reduce our carbon footprint.
"As an alternative route to reducing our carbon footprint we are researching better catalyst materials that can produce and utilise clean fuels that do not pollute our planet."Professor Yan Jiao
Associate Professor Yan Jiao is Director of Research at the University of Adelaide's School of Chemical Engineering and Advanced Materials. By examining how catalysts behave, her team is finding viable ways to produce alternative chemicals and fuels from carbon dioxide and monoxide.
"The world's future energy needs are likely to be met by a mix of renewable sources including alternative liquid fuels which have the advantage that they can be delivered and utilised using existing technology," said Professor Jiao.
Professor Jiao's team works in the field of computational electrochemistry and the design of energy materials by computation methods.
"Generated renewable electricity can lead to both short-term and long-term benefits for our society but the current bottleneck is the conversion and storage of renewable electricity," she said.
"As an alternative route to reducing our carbon footprint we are researching better catalyst materials that can produce and utilise clean fuels that do not pollute our planet.
"We have found that a confined reaction environment created by copper atoms arranged into pyramid-like structures on a nanoscale can selectively transform carbon dioxide and carbon monoxide into ethylene glycol."
The pyramid layout of the atoms is crucial to the copper acting as an effective catalyst in assisting the transformation.
As well as identifying a new reaction mechanism for producing a valuable diol with varied industrial applications, the team's work highlights the potential of designing reaction environments to increase catalyst selectivity and efficiency.
Professor Jiao and Ling Chen, who is a Higher Degree by Research candidate, were recently interviewed about their research by Chemistry World.
"Electrocatalytic conversion of carbon dioxide to chemicals and fuels is a promising path for meeting the carbon-neutral goal advocated by the Paris Agreement on climate change," said Mr Chen.
"Its successful implementation, however, is contingent upon the development of high-selectivity and energy-efficient catalysts.
"Carbon dioxide can be converted electrochemically into single and multi-carbon products. But producing alcohols is more challenging than forming hydrocarbons, and selectively producing higher-value diatomic C2 chemicals such as ethylene glycol remains elusive."
"To the best of our knowledge, a complete route for electrosynthesis of diols such as ethylene glycol, from carbon monoxide and carbon dioxide has never been reported before either experimentally or theoretically."
The team's findings have been reported in the journal Chemical Science.






