A large proportion of the Chinese population suffers from liver disease. Chronic liver fibrosis tends to contribute to cirrhosis, one of the leading causes of death all over the world. At present, there is still a shortage of effective therapies for patients with liver fibrosis and terminal cirrhosis except liver transplantation.
Recently, the journal of Blood published the latest findings of the research team led by Prof. WANG Fudi, Prof. Min Junxia and Prof. ZHENG Shusen. Researchers pioneer in generating hepatocyte-specific transferrin (Trf) knockout mice (Trf-LKO), which sheds light on the molecular mechanism by which Trf deficiencies will lead to ferroptosis-induced liver fibrosis. This research is also the first to identify Slc39a14 as the hepatic transporter of non-Trf-bound iron (NTBI), thereby unraveling a mystery that has baffled academia for over half a century.
Meanwhile, this research also reveals that treating Trf-LKO mice with the ferroptosis inhibitor or deleting Slc39a14 can successfully prevent or attenuate iron overload-induced ferroptosis, thus providing a potential therapeutic target for diagnosing and treating ferroptosis-induced liver fibrosis.
Iron overload in liver tissues has long been found to be intimately bound up with liver dysfunction, but whether it exerts a direct effect on liver damage and how it occurs have remained elusive. As early as 2017, the research team led by Prof. WANG Fudi and Prof. MIN Junxia conducted pioneering research into the pathogenic mechanism in iron overload-induced liver damage by using multiple iron overload knockout mice.
Trf is a serum-abundant metal-binding protein primarily synthesized in the liver. In blood circulation, it binds to ferric ion in a soluble, non-toxic form in order to deliver iron to the bone marrow and other tissues. Under normal conditions, the majority of Trf-bound iron (TBI) is delivered to the bone marrow, and the bulk of NTBI, which can be toxic, is cleared by the liver. The increasing level of NTBI will result in an accumulation of NTBI in hepatocytes.
When the liver lacks Trf, why is NTBI still able to enter it and add up in it, therefore causing cell toxicity? In addition to Trf, is there any other matter which functions as a "porter"? Scientists have been bewildered by these questions for long.
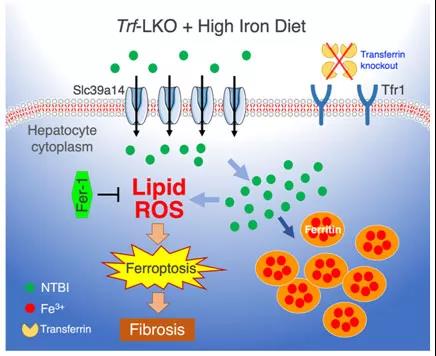
In this study, the research team successfully fabricates hepatocyte-specific Trf-LKO mice, which are viable and fertile but have impaired erythropoiesis and altered iron metabolisms. Researchers are amazed to find that treating Trf-LKO mice with the ferroptosis inhibitor ferrostatin-1 can effectively rescue liver fibrosis induced by either high dietary iron or carbon tetrachloride injections. In addition, deleting hepatic Slc39a14 expression in Trf-LKO mice can significantly diminish hepatic iron accumulation, thereby reducing ferroptosis-mediated liver fibrosis.
Taken together, this research reveals that hepatic transferrin plays a protective role in maintaining liver functions and opens up a promising therapeutic target in treating ferroptosis-induced liver fibrosis.






