decades before memory loss or other symptoms appear - according to new research from Mayo Clinic.
The study, published in Alzheimer's & Dementia: The Journal of the Alzheimer's Association, maps when key brain and blood-based changes tend to accelerate across the lifespan, offering new insight into when detection and prevention efforts could have the greatest impact.
Alzheimer's disease, the most common form of dementia, affects about 6.9 million people in the U.S. aged 65 and older. It involves abnormal changes in proteins such as amyloid and tau that can begin years before symptoms, and it is associated with cognitive decline. There currently is no cure.
Mayo Clinic researchers identified when these changes tend to occur throughout the lifespan. Earlier detection can give patients and families more time to plan, to access care and to benefit from treatments that may slow progression.

Using data from 2,082 participants in the long-running Mayo Clinic Study of Aging, researchers analyzed a wide range of measures - including blood biomarkers, brain imaging and cognitive performance - to identify when Alzheimer's-related changes begin to speed up.
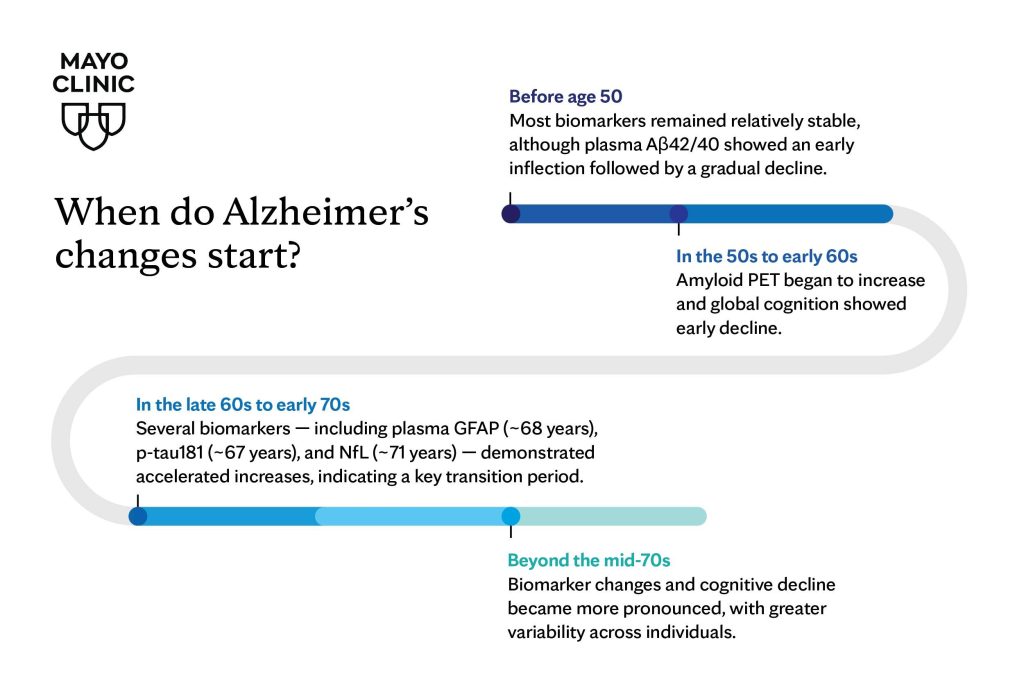
"This population-based study provides an integrated view of age-related patterns across multiple Alzheimer's biomarkers measured in blood and imaging, plus cognition," says Mingzhao Hu, Ph.D., assistant professor in Mayo Clinic's Department of Quantitative Health Sciences and first author of the study. "By estimating the ages when changes in health markers become more noticeable, the results show that many of these shifts tend to happen from late 50s through early 70s."
The future of detecting Alzheimer's

"As Alzheimer's research shifts toward prevention and earlier treatment, blood biomarkers will play a central role in identifying who is best suited for these therapies," says Jonathan Graff-Radford, M.D., chair of Behavioral Neurology at Mayo Clinic and senior author of the study. "Knowing when these biomarkers start to change, and when they relate to cognitive impairment, helps point us to the ages when prevention screening could have the greatest impact."
Researchers found that many Alzheimer's-related biomarkers show that changes begin to accelerate at specific ages.
Measurable declines in cognitive performance were seen to accelerate in people who are in their late 50s, followed by more rapid amyloid accumulation in the brains of people in their early 60s - pointing to an early 60s window when cognitive and amyloid changes become more pronounced. The buildup of amyloid-beta proteins that clump together to form plaques in the brain is a primary hallmark of the disease.
By the late 60s to early 70s, biomarkers of tau pathology and neurodegeneration show more pronounced increases. Several blood-based markers - including plasma GFAP, NfL and p-tau - show steeper changes around ages 68 to 72, alongside more evident brain atrophy, particularly in memory-related regions. Two broad windows emerged, around the early 60s for cognition and amyloid PET, and around the late 60s to early 70s for several blood and neurodegeneration markers, highlighting these key transitional periods in the aging process.
Moving toward earlier detection
Understanding the timeline of Alzheimer's disease progression could be critical for shifting care from late-stage treatment to earlier detection and prevention. Researchers point out that the findings reflect overall population trends, rather than precise predictions for any one individual. However, they offer direction for future research, including examining whether these "breakpoints" can predict cognitive decline, confirming the results in more diverse populations and tracking individuals over time to better understand how the disease progresses.
The results of the study also reinforce the growing role of blood tests in Alzheimer's research and care. These tests showed patterns similar to brain imaging, suggesting they could be used to monitor disease-related changes over time and identify people at higher risk.
"When you think about population screening, the critical issue is timing," says Dr. Graff‑Radford. "You don't want to start too early, before biomarkers change, and this work provides a way to begin addressing that," adds Dr. Graff-Radford.
The work also helps inform research into screening and monitoring by identifying age ranges when blood tests may be most informative. Additionally, several of the blood marker patterns were consistent across two commonly used laboratory platforms, supporting that the findings are not tied to a single assay.
This research is part of a broader effort at Mayo Clinic known as the Precure initiative. It focuses on developing tools to help clinicians detect and address disease-related changes earlier, before symptoms appear or conditions become harder to treat.
For a complete list of authors, disclosures and funding, review the study.






