Certain immune cells can cause damage to the aging central nervous system, according to a novel study by scientists of the University Hospital and the University of Würzburg.
Aging is the biggest risk factor for perturbation of the nervous system, even in the absence of distinct disease or trauma. For yet unknown reasons, the impulse conducting, myelinated projections and synaptic connections between nerve cells are especially vulnerable to aging-related degeneration. These pathological alterations often manifest as cognitive, sensory, and motor decline in older adults and represent a serious socio-economic challenge.
Malactivation leads to damage
Scientists have long assumed that inflammation plays an important role in this process. Mal- or overactivation of distinct cells belonging to the innate immune system - the microglia - appears to promote damage of nerve fibers and synapses in the aging central nervous system (CNS). In a recent project, scientists of the University Hospital Würzburg have now discovered an important role of the adaptive immune system.
The study was conducted at the Department of Neurology under corresponding author and lecturer Dr. Janos Groh from the section of "Developmental Neurobiology" (Prof. Dr. Rudolf Martini) in collaboration with Professor Wolfgang Kastenmüller (Institute for Systems Immunology) and Dr. Antoine-Emmanuel Saliba from the Helmholtz Institute for RNA-based Infection Research. The results of the study have now been published in the scientific journal Nature Aging.
T cells as mediators of neurodegeneration
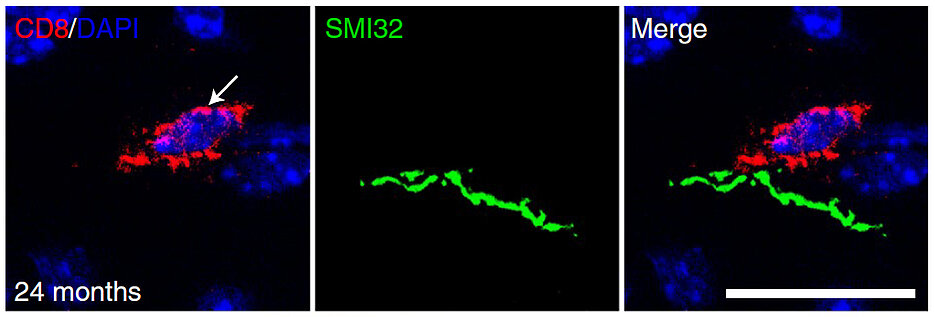
"Cytotoxic CD8+ T cells normally recognize and fight infected or malignant cells. However, in case of autoimmune diseases like multiple sclerosis, they can also do unwanted damage in the nervous system", says Janos Groh. The scientists could previously identify such CD8+ T cells as important disease amplifiers in models of various genetically mediated neurological disorders. Their role in the aging CNS, however, was so far poorly investigated on a functional level. In order to shed light on this question, the researchers investigated the impact of CD8+ T cells in aged mice, where these cells were found in increased numbers in fiber tracts.
"We show that the accumulation of CD8+ T cells leads to degeneration of nerve fibers in the CNS of normal aging mice, which contributes to motor and cognitive decline", Groh summarizes the most important results of the study. Using modern gene expression analyses on the single-cell level, the team could for the first time characterize distinct populations of these CD8+ T cells in the brains of adult and aged mice in detail. This helped the scientists to subsequently clarify how the CD8+ T cells cause harm in the brain using precise immunological animal experiments.
Inflammation as a risk as well as therapeutic opportunity
"In addition, we show that T cell-mediated damage in aged but not adult mice is aggravated by systemic inflammation", Groh adds. According to him, the study therefore confirms that CD8+ T cells are important effectors of inflammation-driven damage to the aging CNS, for example also after infections at more distant sites of the body. In future studies, the researchers want to clarify why and how exactly this inflammatory response is initiated.
Finally, the scientists could find very similar T cell reactions as observed in mice also in autopsies of CNS white matter from older humans. CD8+ T cells might therefore represent a putative target for therapeutic approaches to mitigate aging-related decline of structure and function of the nervous system. The study thus provides basic-scientific and translationally relevant insights into degenerative aging-related processes and another example for the complex interaction between the nervous and the immune system.
Original publication
Accumulation of cytotoxic T cells in the aged CNS leads to axon degeneration and contributes to cognitive and motor decline. Janos Groh, Konrad Knöpper, Panagiota Arampatzi, Xidi Yuan, Lena Lößlein, Antoine-Emmanuel Saliba, Wolfgang Kastenmüller & Rudolf Martini. Nat Aging 1, 357-367 (2021). https://doi.org/10.1038/s43587-021-00049-z






