This article is an excerpt from the research magazine "Tackling Global Issues vol.2 New Era of Radiation Therapy to Fight Cancer." Click here to see the table of contents.
Shinichi Shimizu is relentlessly pursuing research to elevate Hokkaido University to one of the world's best institutions in cancer treatment. His goal now is to allow medical professionals to both plan and conduct therapies right in the treatment room with the help of improved computed tomography.

Dr. Shinichi Shimizu, Professor of Radiation Medical Science and Engineering at Hokkaido University and specialist of prostate cancer radiation therapy.
His research areas include clinical internal medicine and radiation science.
Professor Shinichi Shimizu was dispatched to the University of Texas MD Anderson Cancer Center for three months in 2012 to gain first-hand experience with proton therapy in anticipation of the establishment of Hokkaido University's Proton Beam Therapy Center in 2014. Now, several highly-rated cancer treatment centers in the U.S. have already or are considering adopting a proton therapy device developed jointly by Hokkaido University and Hitachi, Ltd. for its superior performance.
 "We can safely say that the system jointly developed by Hokkaido University and Hitachi is one of the best in the world," Shimizu said. With his expertise in radiation medicine and computer programing, Shimizu played a pivotal role in developing the four-dimensional tumor tracking system, which is called the real-time-image gated proton therapy, resulting in 43 patents in Japan and 20 abroad. Yet he is by no means satisfied with the system and is trying to improve it further through ongoing research on Real-world Adaptive Proton Beam Therapy (RAPT).
"We can safely say that the system jointly developed by Hokkaido University and Hitachi is one of the best in the world," Shimizu said. With his expertise in radiation medicine and computer programing, Shimizu played a pivotal role in developing the four-dimensional tumor tracking system, which is called the real-time-image gated proton therapy, resulting in 43 patents in Japan and 20 abroad. Yet he is by no means satisfied with the system and is trying to improve it further through ongoing research on Real-world Adaptive Proton Beam Therapy (RAPT).
One of the daunting challenges in proton therapy is pinpointing the exact location and shape of moving tumors. Tumors move with respiratory movements and other bodily motions, and their shape and location during a radiation treatment may differ from what they were before, making precise dose delivery difficult. "There is still a certain level of uncertainty in proton therapy because we can't actually see the tumor during the treatment," Shimizu said. "The exact shape and location of a tumor can be different from what is in CT images taken a day before."
"Hokkaido University has specialized in high-precision four-dimensional tumor tracking, but there is more room for improvement. With the help of a gold marker implanted near the tumor, we aim to more accurately visualize the tumor during treatment."
Reducing "artifacts": a vital next step
The RAPT system is designed to obtain clear, reconstructed CT images of a tumor by using only images when the gold marker is in a certain position. It also seeks to improve the quality of tumor images by removing "artifacts," which are distortions or fuzziness in images caused by respiratory movements or the presence of metals in the body. Metals, such as dental fillings and implants, can cause lines to appear in an image, which makes identifying the boundary between a tumor and healthy tissues and vital organs more difficult.
"If we have clear images, we can reduce the area of proton beam irradiation and increase the dose the tumor receives to more effectively fight the cancer while further reducing side effects," Shimizu explains.
In the RAPT system, cone beam computed tomography (CBCT), which uses X-rays coming from two directions, is used in the treatment room with the patient lying on the bed. The CBCT device rotates 360 degrees around the patient while irradiating X-rays from two directions, gathering a massive amount of data on the diseased area, with a gold marker inserted near the tumor serving as a reference point.
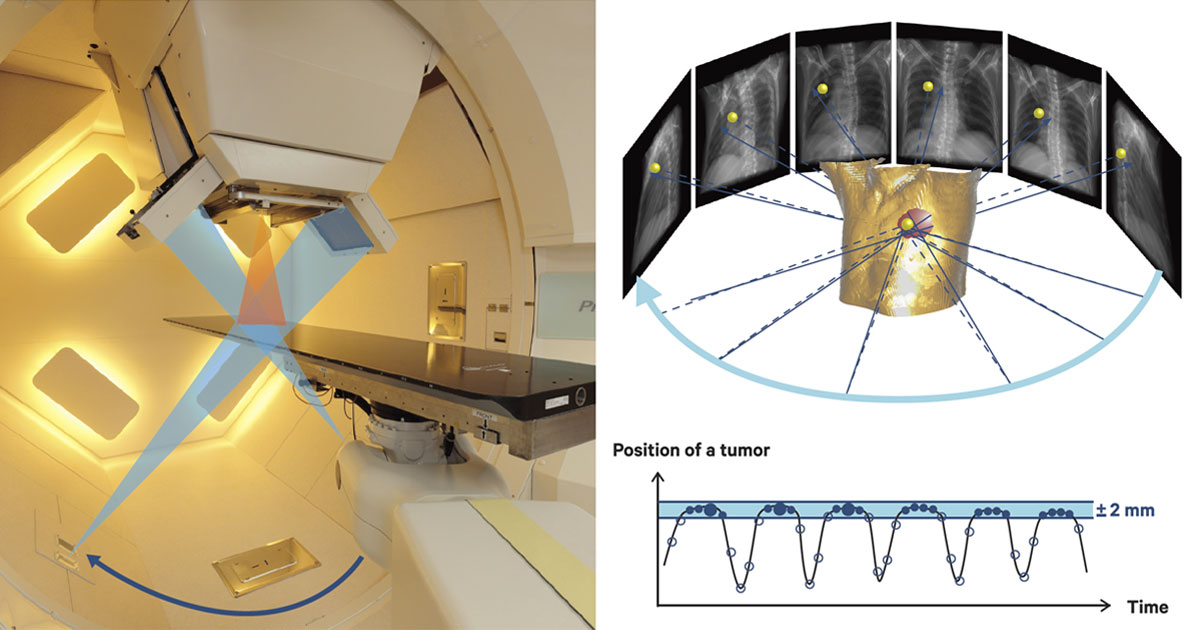
Imaging procedure of the cone beam computed tomography (CBCT). The patient is exposed to X-ray beams emitting from two axes in order to obtain images of the tumor. Images taken when the gold marker is within the 2 mm-gating window are selected to improve image quality.
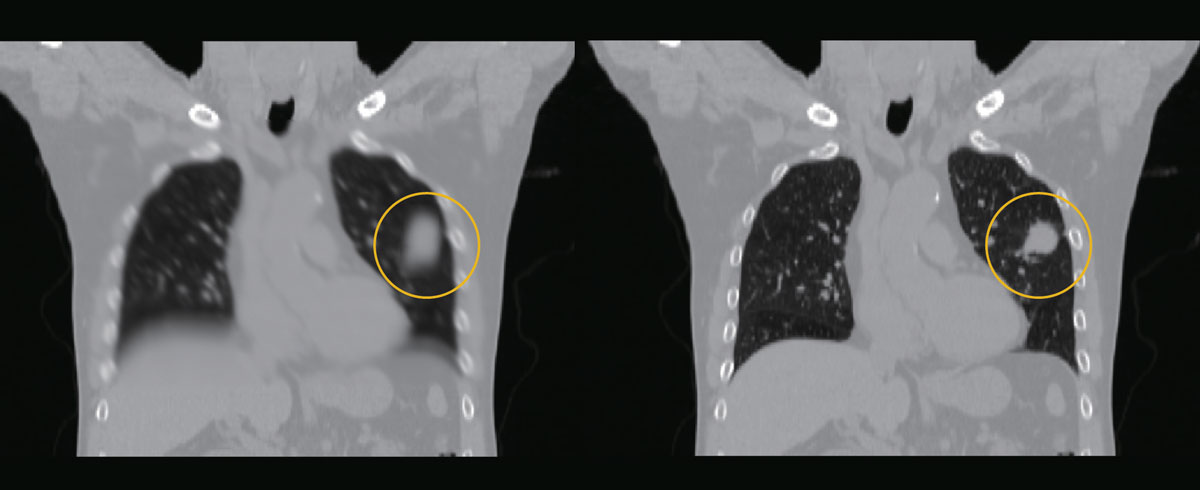
An ordinary reconstruction of a tumor image (left), and a four-dimensional reconstruction of a tumor image made with CBCT images when a gold marker is in a certain position (right).
Hastening treatment decisions
 The position of the gold marker is determined by projection images obtained by CBCT. Only the images in which the gold marker is in a certain position - the gating range - will be used for the four-dimensional reconstruction of the tumor. This will then be used to plan the patient's proton therapy. "Currently, we take CT images before the treatment to plan proton therapy, but in the future with the new system we will be able to do it right in the treatment room," he said.
The position of the gold marker is determined by projection images obtained by CBCT. Only the images in which the gold marker is in a certain position - the gating range - will be used for the four-dimensional reconstruction of the tumor. This will then be used to plan the patient's proton therapy. "Currently, we take CT images before the treatment to plan proton therapy, but in the future with the new system we will be able to do it right in the treatment room," he said.
However, a formidable challenge remains before the treatment can become a reality.
Currently, it takes several hours to identify the range of a tumor's border with healthy tissue, based on images taken before the treatment and elaborate computer calculations. Shimizu says that eventually this task will need to be done automatically by artificial intelligence (AI) for the RAPT system to be usable in the treatment room. However, no such technology yet exists. "We will probably end up using technology that somebody else develops because that in itself is a good research topic," Shimizu said. "We are trying to clear hurdles one by one to make this kind of treatment possible in the future." Shimizu is also conducting research to determine whether tumor tracking is possible without the use of a gold marker, which would make the treatment less invasive.
Besides his own research, Shimizu is busy treating patients at the Proton Beam Therapy Center. As a specialist in prostate cancer radiation therapy, Shimizu says he is determined to do the best for every patient who comes to Hokkaido University Hospital. For some, the center is their last resort. "We have treated cancer patients who were declared incurable by other hospitals," he said. "Some were cured. Some were not. But we did our best for all of them."
Shimizu admitted that his research will not generate as much public attention as cancer immunotherapy, which became a hot topic after two scientists were awarded the 2018 Nobel Prize in Physiology or Medicine for their research in that field. "We are simply trying to improve what we have done in proton therapy," Shimizu said. "Doing so may not lead to a major scientific breakthrough, but we will continue to go forward with our research to make proton therapy a better treatment option for cancer patients and improve their post-treatment quality of life."
Click here to see the table of contents.







