What the research is about
When you catch a cold, an invisible battle takes place inside your body. In this battle, molecules called antibodies bind tightly to invading viruses and bacteria.
Antibodies have a shape similar to the letter Y. The tips of the two arms act like sensors, recognizing and binding to invading pathogens. The lower part of the Y acts like a transmitter, sending signals that alert immune cells: "An enemy has been found!" Guided by this signal, immune cells locate and attack the invaders.
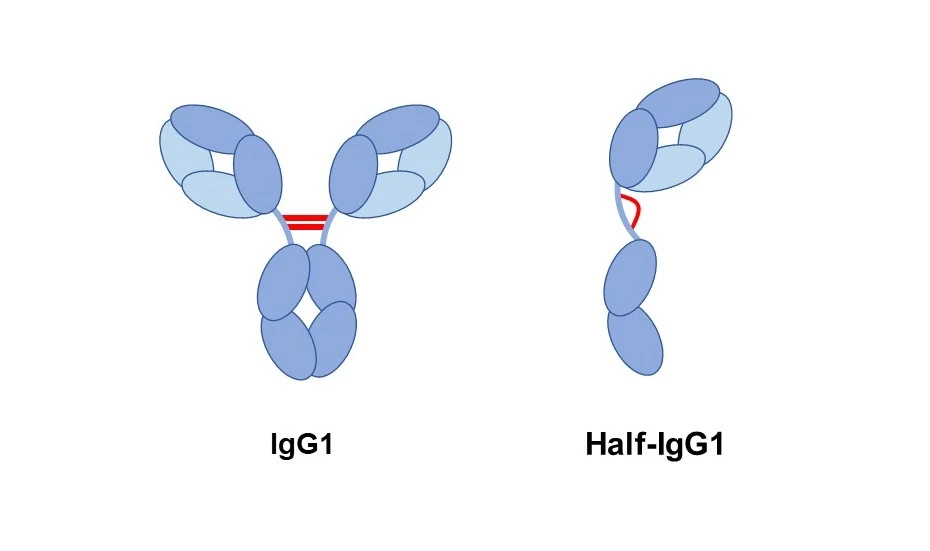
Among many types of antibodies, IgG1 is the most abundant in the human body and is widely used in medical treatments. IgG1 has long been believed to function only when two identical parts join together back-to-back to form a stable pair. The region connecting the arms (sensors) and the lower part (transmitter) - a flexible "waist" of the molecule - allows the antibody to move freely and efficiently capture pathogens.
However, the detailed structure of this waist region has remained unclear. Because it is constantly moving, it has been difficult to observe its shape precisely. Yet scientists suspected that this region might contain an important site that helps connect the two parts of the antibody.
A research team led by Associate Professor Saeko Yanaka at Institute of Science Tokyo (Science Tokyo) focused on a single amino acid in this region that could strongly influence the antibody's shape.
Why this matters
When the researchers removed just this one amino acid, something surprising happened. Normally, two parts of IgG1 join together to form a Y-shaped antibody. But in this case, each part changed its shape and became stable on its own, existing as a single unit rather than a pair. The team named this new form "half IgG1" (Figure 1).
Even more surprisingly, this half antibody was not incomplete. Although it consisted of only one part, it could still recognize pathogens. Moreover, it was able to send signals to a specific type of immune cell, showing a unique and selective function. This finding suggests that the molecule was not simply "broken in half." Instead, it represents a new type of antibody specialized for a particular role.
What's next
This discovery may challenge the long-standing assumption that antibodies must always work as pairs. By taking advantage of the half antibody's ability to interact selectively with certain immune receptors, scientists may be able to design new antibody-based medicines with fewer side effects.
The findings may also provide researchers with a valuable tool for studying the detailed mechanisms of immune responses.
Comment from the researcher
At the atomic level, the difference was only a single amino acid. Yet this tiny change dramatically altered the motion and properties of the entire molecule. By continuing to question a part of the molecule that had long been overlooked, we were able to make this discovery. It reminds us that small molecular changes inside the body can add up to produce powerful functions and new possibilities.
(Saeko Yanaka, Associate Professor, Materials and Structures Laboratory, Institute of Integrated Research, Institute of Science Tokyo)










