Researchers in the lab of Harvard Medical School structural biologist Hao Wu have deciphered the step-by-step assembly of a molecular complex that's crucial for fighting off infections but can also give rise to myriad diseases involving inflammation, from gout to Alzheimer's to the cytokine storms of severe COVID-19.
- By STEPHANIE DUTCHEN
The findings, published in Nature on Nov. 28, forge a path for researchers to develop drugs that regulate the complex's formation and activity to treat or prevent the diseases it's linked to.
When viruses or bacteria enter the body, NLRP3 molecules inside white blood cells known as macrophages detect cell damage caused by the invaders and spring into action.
The molecules then somehow link with other proteins to form inflammasomes: disk-shaped structures that sound an alarm for the immune system to attack and tell infected or otherwise damaged cells to self-destruct.
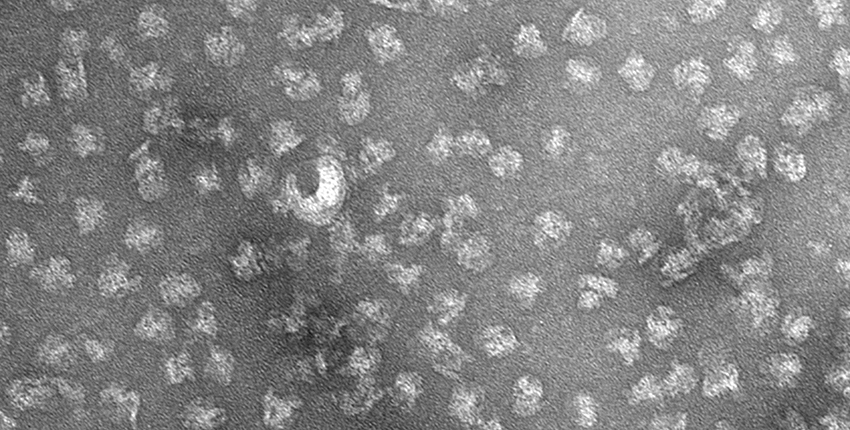
After years of painstaking work, in 2019 Wu's group revealed the precise molecular structure of NLRP3 in its inactive form and took a crack at predicting the steps that lead to inflammasome assembly. Now the team has completed the picture by detailing how NLRP3 morphs into its active form and revealing precisely how 10 of these molecules link together and recruit the other proteins to form the inflammasome.
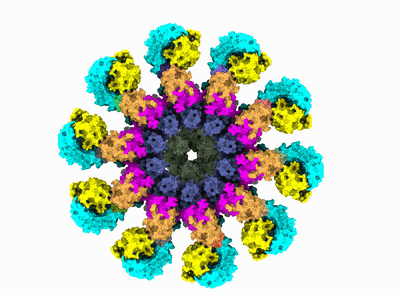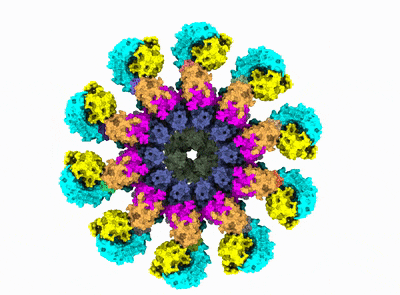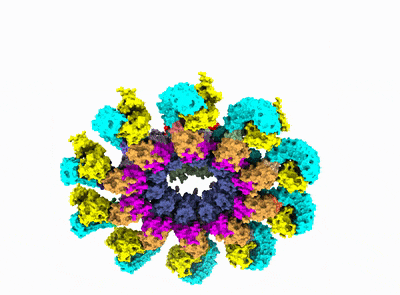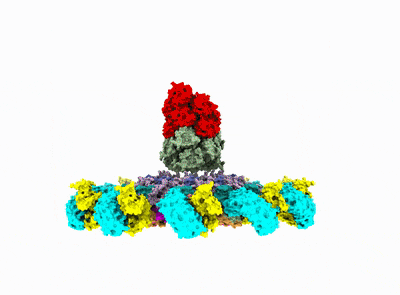
"This is the exciting culmination of a project that many generations of lab members worked on before me," said the study's first author, Le Xiao, research fellow in biological chemistry and molecular pharmacology in the Wu lab.
The achievement was made possible by cryo-electron microscopy. The 3D images in near-atomic resolution represent the first time anyone has seen the inflammasome in its active state.
The findings are important not only for illuminating healthy immune responses but also for understanding how the plot takes a sinister turn in a range of inflammatory diseases.
Due to mutations or other factors, NLRP3 may overreact and form inflammasomes in response not to harmful invaders but to naturally occurring substances in the body - arterial plaques, uric acid crystals, or even healthy tissues. That can drive autoimmune diseases; chronic inflammation that contributes to cardiovascular disease, type 2 diabetes, gout, and the most severe form of nonalcoholic fatty liver disease; and neuroinflammation associated with conditions such as Alzheimer's and Parkinson's diseases.
NLRP3 sometimes fails to deactivate inflammasomes after they've done their jobs. This can lead to life-threatening conditions such as cytokine storms - a hyperactivation of immune cells seen in some severe cases of COVID-19, for example - and sepsis, a runaway systemic reaction to infection.
Conversely, some cancers interfere with NLRP3's ability to form inflammasomes, which then allows the abnormal cells to persist and divide.
For all these reasons, researchers have been trying for years to identify and develop drugs that act on NLRP3, other components of the inflammasome, or molecules activated by inflammasomes. The Wu group's findings provide critical new insights.
"NLRP3 is one of the hottest targets in drug development, and understanding how the inflammasome forms shows us new ways to intervene," said senior author Wu, professor of biological chemistry and molecular pharmacology in the Blavatnik Institute at HMS.
"We hope the insights in our study will provide new avenues for preventing illness and saving lives," said Xiao.
Wu is also the HMS Asa and Patricia Springer Professor of Structural Biology at Boston Children's Hospital. Venkat Giri Magupalli, formerly of the Wu lab, is co-author of the paper.
This work was supported by the National Institutes of Health (grants R01AI124491 and R21AR079766).
Disclosures: Wu is a co-founder of Ventus Therapeutics, a company that seeks to use biological structures to discover new drugs.






