On June 7, Prof. HUANG Jun from the Zhejiang University Life Sciences Institute and ZHOU Zheng from the CAS Institute of Biophysics co-published a research article entitled "Structural insight into BRCA1-BARD1 complex recruitment to damaged chromatin" in the journal Molecular Cell. It reveals the molecular and structural basis for the recruitment of the BRCA1-BARD1 complex to DNA double-strand breaks (DSBs).
DNA is subjected to various kinds of damage every day and DSBs are one of the perilous DNA lesions that may spawn carcinogenesis or cell death. In mammalian cells, homologous recombination (HR) and non-homologous end joining (NHEJ) function as two main pathways to repair DSBs and ensure genome integrity. HR accurately restores genetic information and suppresses tumorigenesis, as it uses a sister chromatid as a template to guide the repair process. Hence, HR pathway is deployed only in the S and G2 phases of the cell cycle, when the sister chromatid is available. The instability of DNA due to defects with HR may well give rise to a multitude of diseases, including tumor. Breast cancer susceptibility protein 1 (BRCA1) can promote HR, which is of supreme importance to maintain genomic stability.
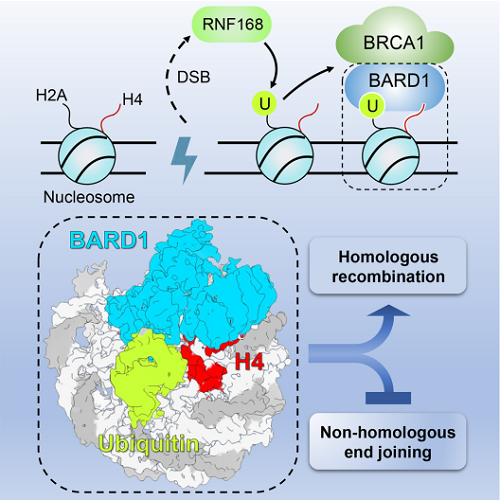
To fulfill its roles in HR, BRCA1 must first be recruited and stably retained at chromatin that flanks DSBs, but how BRCA1 achieves this is not fully understood. Research by ZHOU Zheng and HUANG Jun illustrates how BARD1 reads protein marks in the nucleosomal context to direct the recruitment of the BRCA1-BARD1 complex to DSB sites. Interestingly enough, this study reveals a new ubiquitin (Ub) recognition pattern. To be specific, BARD1 adopts the K63-E64 of ubiquitin (Ub), rather than the common "I44 patch" or "I36 patch" on Ub. It acts as a reader for both DNA damage-induced mark H2AK15ub and DNA replication-associated mark H4K20me0 on the nucleosome. The binding of BARD1 to the ubiquitinated nucleosome core particle (NCPUb) is integral to the recruitment of the BRCA1-BARD1 complex to DSB sites at S/G2 phases. Undermining the BARD1-NCPUb interaction severely impedes the recruitment of the BRCA1-BARD1 complex, contributing to the poorer efficiency of HR and rendering cells more sensitive to ABT-888, a PARP inhibitor (PARPi). This study also identifies multiple cancer-associated BARD1 mutations that disrupt BARD1-NCPUb interactions and hence impair HR.
Together, this study elucidates the mechanism of BRCA1-BARD1 complex recruitment and retention by DSB-flanking nucleosomes and sheds light on cancer therapeutic avenues.






