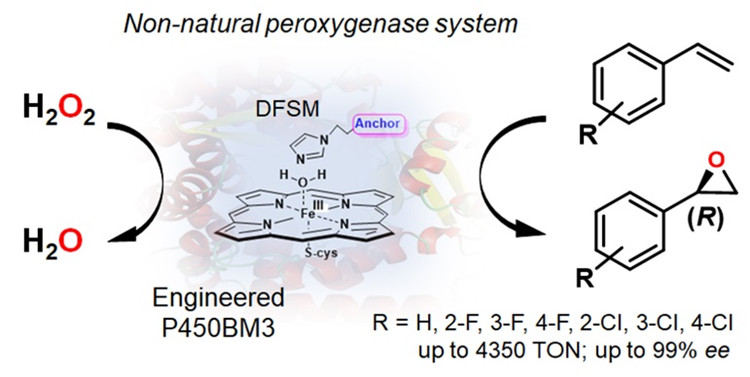
An international team of researchers combined protein engineering and dual-functional small molecule (DFSM) regulation of a unique non-natural P450BM3 peroxygenase to achieve highly H2O2-dependent (R)-enantioselective epoxidation of styrene and its derivatives.
The study was published in Chemical Science, the flagship journal of Royal Society of Chemistry.
"Although in recent years, asymmetric epoxidations gained great development based on synthetic molecular catalysts or biocatalysts. It remains a significant challenge to achieve comparable (R)-enantioselectivity in the epoxidation of unfunctionalized styrene using native enzymes or protein engineering," said Prof. CONG Zhiqi from Qingdao Institute of Bioenergy and Bioprocess Technology (QIBEBT), Chinese Academy of Sciences (CAS).
The researchers constructed a DFSM-facilitated P450 peroxygenase system. This system could not only help to modify enzyme by protein engineering more flexibly, but also apply another fine regulation to substrate cavity by DFSM.
In this new approach, they achieved highly H2O2-dependent (R)-enantioselective epoxidation of styrene and its derivatives. The observed (R)-enantiomeric excess (ee) of styrene epoxidation was up to 99%, and the best mutant (with 98% ee) reached the best activity of P450 towards styrene epoxidation so far.
The study indicates that the synergistic use of protein engineering and an exogenous DFSM is an efficient strategy to control enantioselectivity of styrene epoxidation.