The exact moment a crystal is formed from a previously scattered group of atoms has been captured on video in real time in a world-first experiment by experts in nanotechnology.
The multi-national research team from the UK, Germany and Japan reveal the exact moment of nucleation (formation of a crystal) in a new paper and set of videos published online today in Nature Chemistry.
A huge variety of materials are made of crystals, from ice, table salt and sugar, to more specialised formulations used in medicines, transistors and those found in gemstones. Up to now, exactly how crystals initially form has been an unresolved and contentious issue as it happens at incredibly small scales.
The team's new discovery and capturing of how crystals form in three different types of metal could inform the better design of a range of materials in the future.
Crystal formation at the atomic scale
Nucleation is the very first step in crystal formation, in which a group of atoms or molecules first suddenly stop moving and become ordered in a particular pattern. This process can determine the kind of crystal that ends up forming, which is vitally important as it defines the structure and properties of the material. For example, how the cocoa butter in chocolate crystallises is the difference between the melt-in-your-mouth goodness of a bar of chocolate and the shiny glaze of tempered chocolate used in cooking.

Nano test tubes and electron beams
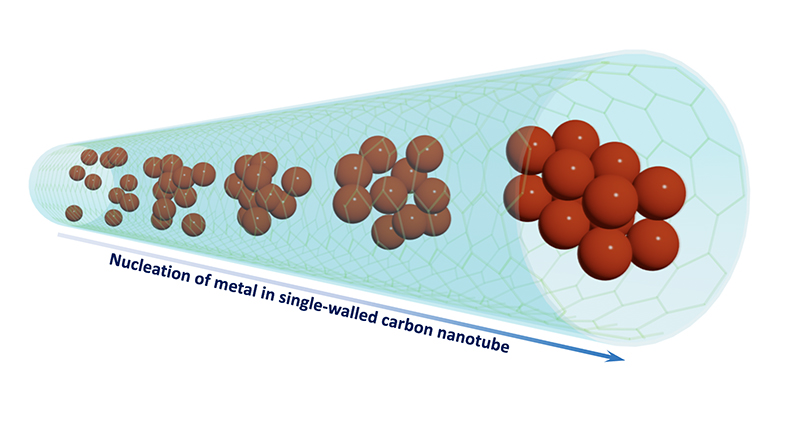
The scientists, led by the University of Nottingham School of Chemistry's Professor Andrei Khlobystov and Professor Ute Kaiser, head of the Electron Microscopy of Materials Science in the University of Ulm, have used carbon nanotubes – atomically thin hollow cylinders of carbon just 1-2 nanometers wide – as miniature test tubes for atoms and molecules to record moving images at the atomic scale using transmission electron microscopy (TEM).
Dr Stephen Skowron, a member of the Nottingham research team who specialises in computational chemistry, said: "To give you a real idea of its size, a carbon nanotube is an atomically thin cylinder with a diameter 80,000 times thinner than a single strand of hair. It gives us a unique opportunity to entrap and study atoms directly by imaging in real-time. As an electron's wavelength is significantly shorter than the size of an atom, the electron beam of TEM allows us to follow the movements of individual metal atoms inside the nanotube with great precision."
The group managed to film the crystal nucleation of three different metals with atomic resolution, in particles composed of only one or two dozen metal atoms. Crucially, in all three cases they were able to record before, during, and after the key moment of nucleation.
Professor Ute Kaiser said: "The direct observation of the entire process of nucleation allowed us to distinguish the point at which the particles were large enough to become crystalline and showed us how many atoms were required for nucleation in our conditions. This was between 10 and 20 atoms for both iron and rhenium, a remarkably small number."

Professor Andrei Khlobystov commented: "The further we develop nanotubes as nanoscale laboratories, the deeper we understand atoms and molecules in action. A combination of nanotubes and electron microscopy imaging reported in this study gives us the most direct experience of atoms and their reactions."
Three metals, three processes, one mechanism
Intriguingly, each metal displayed a different example of how the initially amorphous atomic clusters could overcome the size requirement and energy barrier for nucleation:
- Iron. Fe atoms were individually delivered to an initial seed of only two atoms using an 'atomic injector', until the cluster was large enough to crystallise.
- Gold. A larger amorphous particle of Au was irradiated with the electron beam, providing enough energy for nucleation.
- Rhenium. Two smaller clusters of ~10 Re atoms bumped into each other, merging to form a particle that was large enough to crystallise.
The existence of the initially amorphous clusters was a key to understanding the underlying mechanism of the nucleation processes, as this and the energy barriers to nucleation are requirements for theoretically proposed processes called 'two-step nucleation mechanisms'.
Dr Kecheng Cao, a Research Assistant at Ulm University who performed the experiments said: "Classical nucleation theory predicts crystal formation in single steps by the attachment of atoms to an ordered structure. For all three metals we studied, we found an amorphous precursor and the presence of more than one barrier, consistent with crystallisation via a two-step nucleation mechanism."
Two-step nucleation mechanisms are much more complex and contentious processes compared to classical nucleation theory, with a large degree of scientific debate over both their specific details and whether or not they are required to explain nucleation in a variety of different materials. The research team hopes that by successfully capturing this process in action for the three metals studied, it will shed light on the fundamental phenomenon of nucleation and crystal growth, enabling better-controlled design of materials in the future.
The group of scientists has previously used the same approach to study how chemical bonds form and break, and to film 'movies' of chemical reactions at the single-molecule level.