MARCH 18, 2026, NEW YORK - The immune system's killer T cells do a commendable job of detecting and destroying cancer cells. But the harsh environment at the heart of tumors often saps them of their vitality, pushing them into a state of permanent lassitude called "terminal exhaustion." The phenomenon accounts for why so many tumors resist routine immune clearance and even cancer immunotherapies devised to stimulate their lethal capabilities.
Terminal exhaustion is characterized by an accumulation of dysfunctional mitochondria-the bean-shaped energy generators in cells-and extensive genetic reprogramming that stalls proliferation and hobbles the cell-killing weaponry of T cells. Yet how mitochondrial dysfunction is linked to genetic reprogramming in the cells was unclear. No longer. Researchers led by Ludwig Lausanne's Yingxi Xu and Ping-Chih Ho report in the current issue of Nature how the accumulation of useless mitochondria is linked to T cell exhaustion through a complex series of subcellular processes.
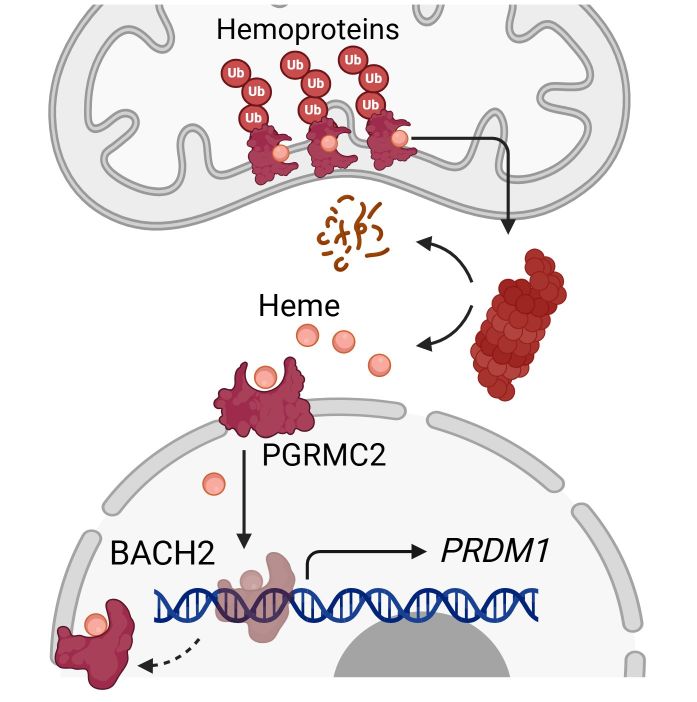
How dysfunctional mitochondria drive T cell exhaustion: Damaged mitochondria in CD8⁺ T cells increase the activity of a protein degrading machine known as the proteasome, which preferentially degrades mitochondrial hemoproteins, releasing "regulatory heme" (RH). Elevated RH travels into the nucleus through the transporter PGRMC2, where it binds to the transcription factor BACH2, inducing its degradation and, consequently, the activation of gene expression programs that push T cells into terminal exhaustion.
"We have long known that exhausted T cells lose their reserves of energy and effectiveness, yet the molecular trigger of this decline was unknown," said Ho. "Our study identifies an unexpected culprit: heme, the ringed, iron-bearing molecule at the heart of hemoglobin that gives blood its red color and is found in many other proteins. We show how the release of heme from dysfunctional mitochondria occurs and how the molecule induces exhaustion. We also demonstrate in preclinical studies that disrupting two discrete steps in these complex processes could significantly enhance the efficacy of T cell-mediated immunotherapies."
In particular, the researchers show how chimeric antigen-receptor (CAR) T cell therapy-in which T cells are taken from a patient, engineered to target a cancer antigen and reinfused to treat their cancer-can be tweaked to improve the vitality and effectiveness of the cells.
Killer T cells that infiltrate tumors to kill cancer cells enter a punishing tumor microenvironment (TME), one that is poor in oxygen and vital nutrients and replete with metabolites that suppress their activity. These challenges cause severe mitochondrial dysfunction.
Normally, useless mitochondria are taken apart, recycled and wholly replaced by fresh young ones. This process, mitophagy, is disrupted in tumor-targeting T cells in the TME, leading to an accumulation of the moth-eaten organelles. Recent evidence indicates this directs the gene expression programs underlying terminal exhaustion. But how exactly that happens was not known.
Xu, Ho and their colleagues report in their paper that the glut of dysfunctional mitochondria enhances the activity of a cellular protein digesting machine, known as the proteasome, in T cells. The activated proteasome, they show, preferentially degrades mitochondrial heme-containing proteins.
As might be expected, this bias leads to quite the buildup of heme in the cells, resulting in the generation of a functionally distinct form of the molecule referred to as "regulatory heme," which zips into the nucleus through a transporter named PGRMC2. There it binds to a transcription factor, a protein that regulates gene expression, causing its degradation. This kicks off a series of events that culminates in the activation of genetic programs known to induce terminal exhaustion.
The researchers show that genetic disruption of PGRMC2 abrogates this effect, keeping anti-tumor T cells in a functionally vibrant state, suggesting it is a potential drug target for the enhancement of T cell-activating cancer immunotherapies.
Xu, Ho and colleagues also examined how the pharmacological inhibition of the proteasome with an existing leukemia therapy, bortezomib, might affect CAR-T cells. Like bortezomib, CAR-T therapy is currently used to treat B cell acute lymphoblastic leukemia (B-ALL).
"We found that the transient and low-dose addition of bortezomib to CAR-T cell cultures during manufacturing reduces exhaustion-associated programs in the cells and induces durable reprogramming of their gene expression patterns to maintain them in a proliferative and functionally vibrant state," said Xu. "We are particularly pleased that an existing drug can be used for that purpose, indicating the strategy might be tested quickly for its efficacy in the clinic."
Notably, in analyzing data from B-ALL patients who received CAR-T cell therapy, the researchers found that high proteasome activity in their CAR-T cells correlated with poor outcomes, while low activity was associated with the opposite, including complete responses. This supports the clinical relevance of their findings in preclinical models.
"Our work reframes T cell exhaustion as being, in part, the product of a reversible imbalance in metabolic signaling within the cell," said Xu. "This insight opens new avenues to the development of next-generation cellular therapies for cancer."
This research was supported by the Ludwig Institute for Cancer Research, the Swiss National Science Foundation, the Cancer Research Institute, Helmut Horten Stiftung, Swiss Cancer Foundation, the Melanoma Research Alliance, the National Key Research and Development Program of China, CAMS Innovation Fund for Medical Sciences, the National Natural Science Foundation of China, the Ministry of Science and Technology in Taiwan, the National Taiwan University Hospital, the Swiss Cancer League, the Geneva Cancer League, the National Science Foundation of China, the UK Medical Research Council and the Suzhou Municipal Key Laboratory.
Ping-Chih Ho is a member of the Lausanne Branch of the Ludwig Institute for Cancer Research and a full professor at the University of Lausanne.






