Gyrification has been observed for the first time in brain organoids grown in vitro using 3D-printed scaffolds.
The research, by an international team from the Autonomous University of Madrid and the Technical University of Denmark, used 3D printing to create scaffolds for engineered flat brain organoids. The scaffolds allowed the brain organoid size to be significantly increased and after 20 days, self-generated folding was observed. Their results are published in the IOP Publishing journal Biofabrication.
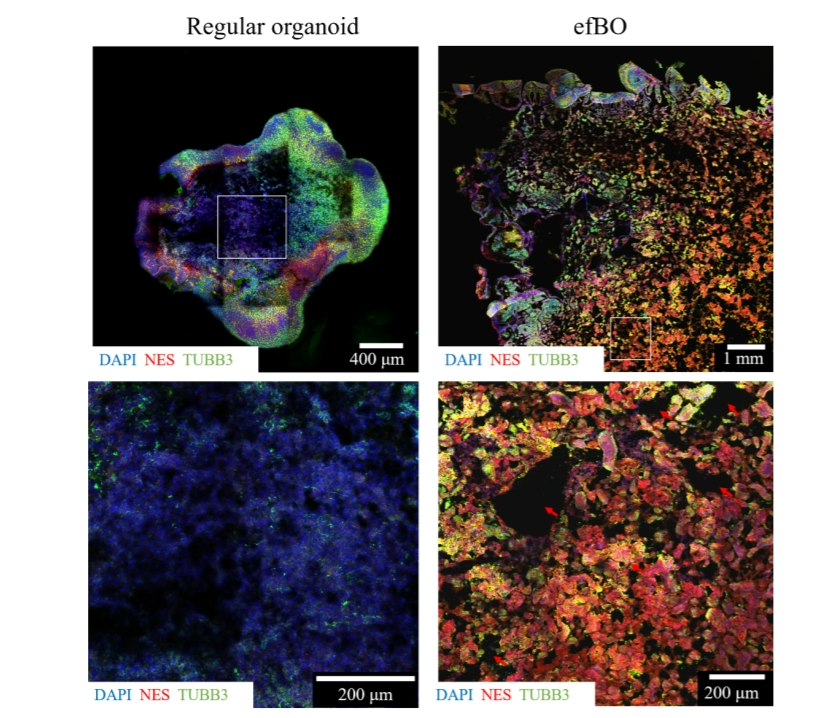
The work aims to address several of the shortcomings of existing brain organoids. One of the lead authors, Theresa Rothenbücher, said: "The lack of vascularization leads to diffusion limitations for nutrients and oxygen, resulting in a necrotic tissue core for organoids larger than approximately 500 μm. In an attempt to solve this problem, brain organoids have been vascularized. While including endothelial cells in the culture system increases the complexity of the model, the generated vessel structures show no functionality (blood flow) in vitro. We are able to circumvent this issue by applying bioengineering techniques."
Another lead author, Hakan Gürbüz, commented: "By culturing brain organoids with a polycaprolactone (PCL) scaffold, we were able to modify their shape into a flat morphology. Engineered Flat Brain Organoids (efBOs) possess advantageous diffusion conditions and thus their tissue is better supplied with oxygen and nutrients, preventing the formation of a necrotic tissue core. The shift from a spherical to a flat shape leads to a significant increase in size and surface-to-volume ratio of the brain organoids." eFBOs also offer increased potential to create biologically relevant systems, due to the complexity of the models that they enable. Ensuring the long-term viability of these models is a major aim of this branch of research, which has been difficult until now; flat organoids address the problem of longevity by avoiding the formation of necrotic tissue.
"The 3D printing of scaffolds was key to overcoming the shape limitations of the previous spherical models. Contributing author Jenny Emneus says, 3D printing enables: "Reproducible fabrication of specific 3D scaffolds with high architectural complexity, precision and design versatility. By introducing a 3D-printed scaffold into the culture protocol, the size of the brain organoids and the tissue density and thickness can be tuned."
The resulting model showed consistent formation of neuroepithelial folding resembling gyrification. Contributing author Alberto Martinez-Serrano said: "…we were able to observe folding reminiscent of gyrification around day 20, which was self-generated by the tissue. To our knowledge, this is the first study that reports intrinsically caused gyrification of neuronal tissue in vitro." The appearance of gyrification reflects a further increase of the surface area and resembles the process of human brain development.
Although brain organoids do not reproduce the exact anatomy of a human brain, they are an important step towards recapitulating the human brain; tissue-equivalent models such as these organoids can replace the use of animal models in research into drug screening for toxicity and the understanding of disease progression. Alberto Martinez-Serrano continued: "The human brain is the most complex organ of the body and due to its inaccessibility, we still lack scientific knowledge about brain development and diseases. Studies with animals are ethically restricted and should be minimized. Especially for drug screening applications, a highly reproducible protocol with simple tissue culture steps and consistent output, is required. We consider our efBO protocol as a next step towards the generation of a stable and reliable human brain model for drug screening applications and spatial patterning experiments."






