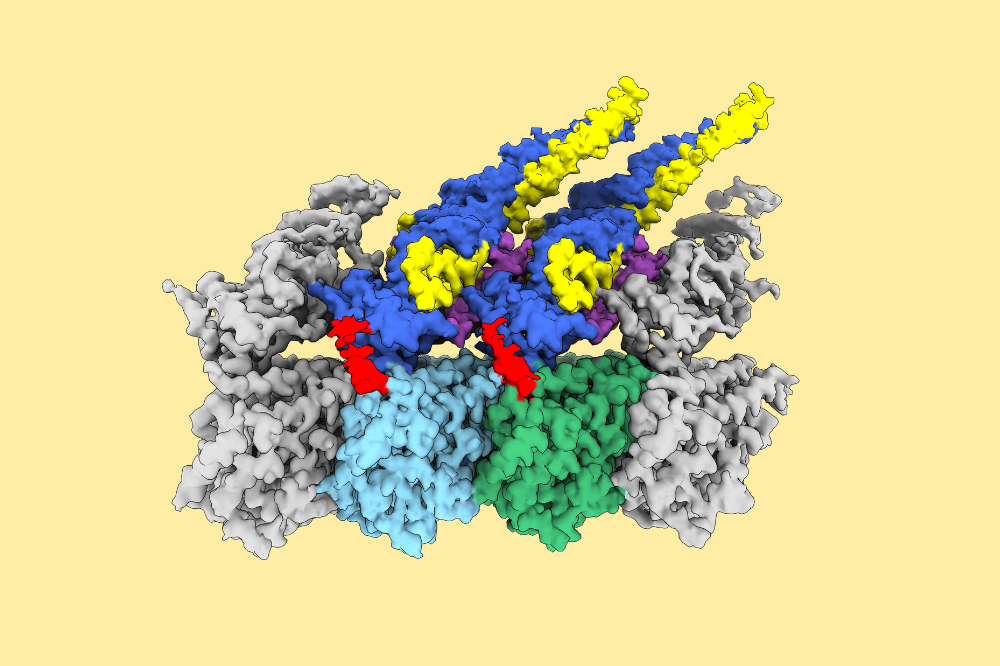
Structure of Ndc80 complex bound to microtubule. Ndc80 complex is the major microtubule3binding module at the kinetochore. Green: α-tubulin, light-blue:β-tubulin, dark blue: HEC1 (a4subunit of the Ndc80 complex), and yellow: NUF2 (a subunit of the Ndc80 complex). (Credit: Funabiki lab)
Microtubules, the dynamic filaments that form the cell's internal scaffolding, have long been viewed as mere passive structural supports. But a new study reveals they play a far more active signaling role. The findings, published in Science Advances, demonstrate that microtubules are in fact regulators of enzymatic reactions through reshaping geometry of the enzyme's substrate proteins attached to them and controlling when key events occur to conduct cell division.
The discovery sheds new light on one of the central challenges of cell division. As chromosomes prepare to separate, they must attach correctly to microtubules so they can be pulled to opposite sides of the cell. When these connections form incorrectly, cells must be able to break the faulty ones without disrupting the correct ones. The study shows that microtubules themselves guide this process, directing the enzyme Aurora B kinase to dismantle bad connections while stabilizing proper ones-a quality-control system that helps prevent chromosome-segregation errors that can lead to abnormalities in the number of chromosomes present, a hallmark of many cancers.
"For a long time, people have regarded microtubules as a structural component of the cytoskeleton," says Yiming Niu, a postdoctoral associate in Hironori Funabiki's Laboratory of Chromosome and Cell Biology at Rockefeller. "Our study illustrates that it is also an active enzyme regulator that does important physiological tasks."
Keeping chromosomes on track
For a cell to divide successfully, it must first copy the chromosomes carrying its genetic material and separate these copies so that each new daughter cell receives a complete set. This delicate task is carried out by the mitotic spindle, a temporary structure of long protein filaments called microtubules that constantly grow and shrink as they probe the cell for chromosomes. Microtubules ultimately latch onto each chromosome at a specialized region called the centromere, where a protein structure known as the kinetochore acts as the chromosome's coupling device. The microtubules pull the chromosome copies toward opposite ends of the cell so that, when the cell divides, each daughter inherits the genetic material that it needs to thrive.
"When the system is working properly, we call it a bi-oriented configuration," Funabiki says. "The kinetochores at the centromeres of each chromosome have attached to the ends of microtubules from opposite sides of the mitotic spindle-they're ready to go."
But the process seldom goes off without a hitch. Microtubules frequently attach incorrectly, sometimes grabbing both sister chromosomes from the same side of the cell or forming tangled connections to multiple spindle poles. Left unchecked, these faulty attachments would cause chromosomes to segregate unevenly during division, producing cells with too many or too few chromosomes. Such errors would lead to chromosomal instability, and a condition found in most cancers, in which cells carry the wrong number of chromosomes. To prevent this, cells rely on a surveillance system that continually tests chromosome attachments, breaking incorrect ones so the cell can try again until the proper configuration is achieved.
At the center of this quality-control system is Aurora B, an enzyme that somehow both destabilizes and stabilizes microtubule connections. During early cell division, Aurora B weakens incorrect chromosome attachments at the kinetochore, allowing them to detach so the cell can try again. At the same time, it suppresses another enzyme called MCAK, a microtubule destroyer that would otherwise chew up the spindle's fibers. Aurora B, in effect, breaks faulty connections while protecting the spindle itself-a balancing act that has long puzzled scientists.
Earlier models suggested that correct chromosome attachments pull kinetochores away from Aurora B at the centromere of each chromosome, reducing the enzyme's ability to modify nearby proteins at the kinetochore. But later evidence showed active Aurora B is found at the kinetochore and can bind directly to microtubules, leading Funabiki's team to propose an alternative. Perhaps microtubules were physically blocking Aurora B from reaching its targets once a chromosome is properly attached.
If proven correct, the idea could reveal how cells normally prevent chromosome mistakes, and how those safeguards break down in cancer. "If you look at cancer, the vast majority of the tumors have an abnormal number of chromosomes," Funabiki says. "It has been suggested that one of the things that causes cancer cells to develop is that this process of correcting misplaced microtubules stops functioning properly."
A new microtubule hypothesis
To test whether microtubules could control Aurora B's activity, the team recreated the system outside the cell using purified proteins. When they combined Aurora B and its regulatory complex with two microtubule-binding partners-the Ndc80 complex, which anchors chromosomes to the spindle, and MCAK, an enzyme that dismantles microtubules-a clear pattern emerged. When Ndc80 was already attached to a microtubule, Aurora B struggled to modify it, suggesting that the clustered complexes were effectively blocking the enzyme's access. But MCAK behaved very differently, remaining fully accessible to the enzyme even when bound to microtubules. The result suggested that microtubules were not just structural supports, but active participants that control which proteins Aurora B can reach.
To see how this worked at the molecular level, the researchers used cryo-electron microscopy. They observed that, when the Ndc80 complex attaches to microtubules, many Ndc80 molecules gather together into clusters along the filament. This clustered state hides the sites that Aurora B normally modifies from the enzyme. When MCAK binds microtubules, however, the sites that Aurora B modifies remain fully exposed, making it easy for the enzyme to make changes and let MCAK off from microtubules.
"When MCAK binds microtubules, the filament acts like a molecular flytrap, bringing the two microtubule-binding proteins, MCAK and Aurora B, together and making it easy for the enzyme to rapidly dismantle MCAK," Funabiki says.
The result is a kind of molecular switch controlled by microtubules. If Aurora B reaches the Ndc80 complex before clustering occurs, the enzyme weakens the attachment so the cell can break it and try again. But once the microtubule organizes Ndc80 into clusters, Aurora B can no longer reach its targets, and the correct chromosome attachment is locked in place. Experiments in living cells confirmed that microtubules must organize Ndc80 into clusters to maintain stable chromosome attachments. Cells engineered with mutant Ndc80 that could bind microtubules but could not cluster struggled to maintain stable attachments and divide chromosomes evenly.
Together, the findings show that microtubules are not just structural scaffolds during cell division. By reshaping the proteins that bind to them, they control when Aurora B can act, shielding some targets while exposing others and ensuring chromosomes separate with remarkable precision.
"Our work reframes how we think about the role of microtubules inside the cell," Niu says. "We now know that the microtubule actively controls which biochemical reactions occur and when."
The findings also offer the most detailed view yet of how cells stabilize correct chromosome attachments; understanding how cells distinguish correct attachments from faulty ones provides a molecular blueprint for studying how this safeguard fails in disease. "Knowing how cells normally correct these microtubule attachments can help us explain what happens when that process fails," Funabiki says.






