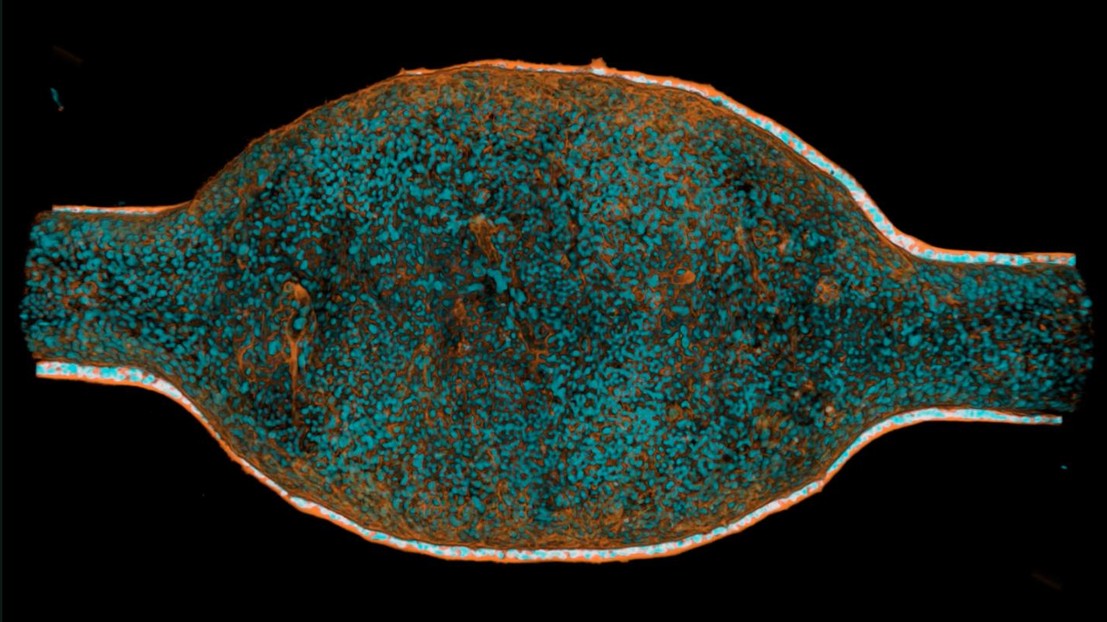
Animated Z-stack showing a volume of a differentiated mini-bladder stained with phalloidin (orange) and DAPI (cyan). Credit: Paduthol et al 2026.
Researchers at EPFL, Heidelberg University and Roche have built a human mini-bladder to show how urine composition weakens bladder tissue, helping infections recur even after antibiotics.
The bladder is not just a static storage bag; its lining stretches, relaxes, and stays in constant contact with urine whose composition changes with hydration, diet, and disease. Urine can vary widely in salt and solute concentration, and studies on animals have shown that concentrated urine can damage the bladder lining. But how exactly that happens is unclear.
UTIs impact over 400 million people worldwide every year. Most recurrent urinary tract infections (UTIs) are caused by a bacterium, "uropathogenic Escherichia coli", or UPEC. These bacteria can live in urine, stick to bladder cells, and invade the bladder tissue. Some forms can even hide inside cells or enter dormant states that make them harder to kill. And so far, there has been no human model able to capture the full interaction between urine, bladder tissue, and bacteria over time.
A custom-made human bladder
Now, scientists at EPFL, Roche's Institute of Human Biology in Basel, and Heidelberg University have developed a human mini-bladder by integrating organoid technology with bioengineering to create a lab-grown model that mimics the organ's complex architecture.
The mini-bladder recreates the layered structure of the lining of the human bladder, exposes it to real or synthetic urine, and mimics filling and emptying cycles, revealing how urine composition shapes tissue health and bacterial survival during infection.
The work was led by John McKinney (EPFL) Matthias Lütolf (Roche Institute of Human Biology/EPFL), and Vivek Thacker (Heidelberg University). It is published in Nature Communications.
Put to the test
The researchers began by growing primary human bladder cells inside a small microfluidic device. Over several weeks, the cells formed a stratified urothelium (the inner lining of the urinary tract) with a tight barrier, similar to the human bladder. The tissue could stretch and relax, mimicking bladder filling and voiding.
The team then perfused the mini-bladder with urine of different but defined compositions. They compared urine with low solute concentration with urine with high solute concentration. They also infected the mini-bladder with UPEC and monitored its effects during repeated wash cycles that simulate urination. They also ran the tests with common antibiotics, including ciprofloxacin and fosfomycin.
High-solute urine gives bacteria an advantage
The study showed that long-term exposure to high-solute urine damages the bladder tissue. Barriers ("tight junctions") weakened, more bladder cells died than in control tissues, and the expression of genes involved in immune defense dropped. In contrast, control mini-bladders, exposed to low-solute urine stayed healthier and more resilient.
"When infected with UPEC, mini-bladders exposed to concentrated urine accumulated more bacteria inside the tissue," says Gauri Paduthol, the study's first author. "These tissue-associated bacteria were harder to clear with antibiotics. Fosfomycin, a widely used, first-line treatment for UTIs, triggered a surprising response: in high-solute urine, UPEC switched into unusual 'cell wall-deficient bacteria' forms that resisted antibiotic treatment."
The study also found that these cell wall-deficient bacteria were not only present in urine; they were also located deep within the bladder tissue, lodged between cells. After the antibiotic was removed, the surviving bacteria reseeded the infection, providing a direct mechanism for recurrence.
The results suggest that urine composition is an active player in urinary tract infections, and that concentrated urine weakens bladder defenses and helps bacteria survive treatment. This may help explain why some patients suffer repeated infections, especially when urine stays highly concentrated for long periods.
In addition, the newly developed human mini-bladder offers a powerful new tool to study UTIs and test therapies under realistic conditions.
Other contributors
- EPFL BioElectron Microscopy Facility
- EPFL Bioinformatics Competence Centre
- University of Lausanne






