A laboratory preclinical model of the most common special histological subtype of breast cancer. Courtesy of George Sflomos (EPFL)
EPFL researchers have developed preclinical models for invasive lobular carcinoma and trialed a new drug. It slows tumor growth effectively by targeting specific features of the disease and opening the door to future clinical trials.
Breast cancer is not a single disease. Some types of carcinoma do not form distinct masses but spread in a string-like pattern, making them difficult to detect. This is the case with invasive lobular carcinomas, or ILCs, where cancer cells detach from each other and grow in thin, diffuse strands. They account for 10 to 15% of breast cancers.
But since clinical trials rely on measuring tumor size to judge whether a treatment is working, these tumors are often not considered, and, as a result, most lobular carcinomapatients receive treatments that have been developed for other breast cancers, and there is no treatment specifically tailored to ILC's unique biology.
"One major reason is that ILC has been poorly represented in clinical trials, and scientists have lacked good laboratory models that faithfully reproduce how this cancer behaves in patients," says Professor Cathrin Brisken at EPFL's Swiss Institute of Cancer Research and the Institute of Cancer Research (UK).
Brisken has now led an international team of scientists in developing and testing a new promising drug specifically for lobular carcinoma. The drug, called PXS-5505, is already being evaluated and shown to be well tolerated in early-phase clinical trials for another disease, myelofibrosis, meaning that its safety is being established, and that can accelerate translation to ILC clinical trials.
More realistic models
"We have solved part of the problem by creating new, more realistic models of lobular carcinoma using a technique that grows human cancer cells inside mouse milk ducts, called intraductal xenografts," she explains.
The models better reproduce how cancer grows and spreads in patients, including hormone receptor status and metastatic behavior. "Our new models allow researchers to study the cancer cells and test treatments under conditions that closely resemble human disease," says Brisken.
The study is published in Cancer Research.
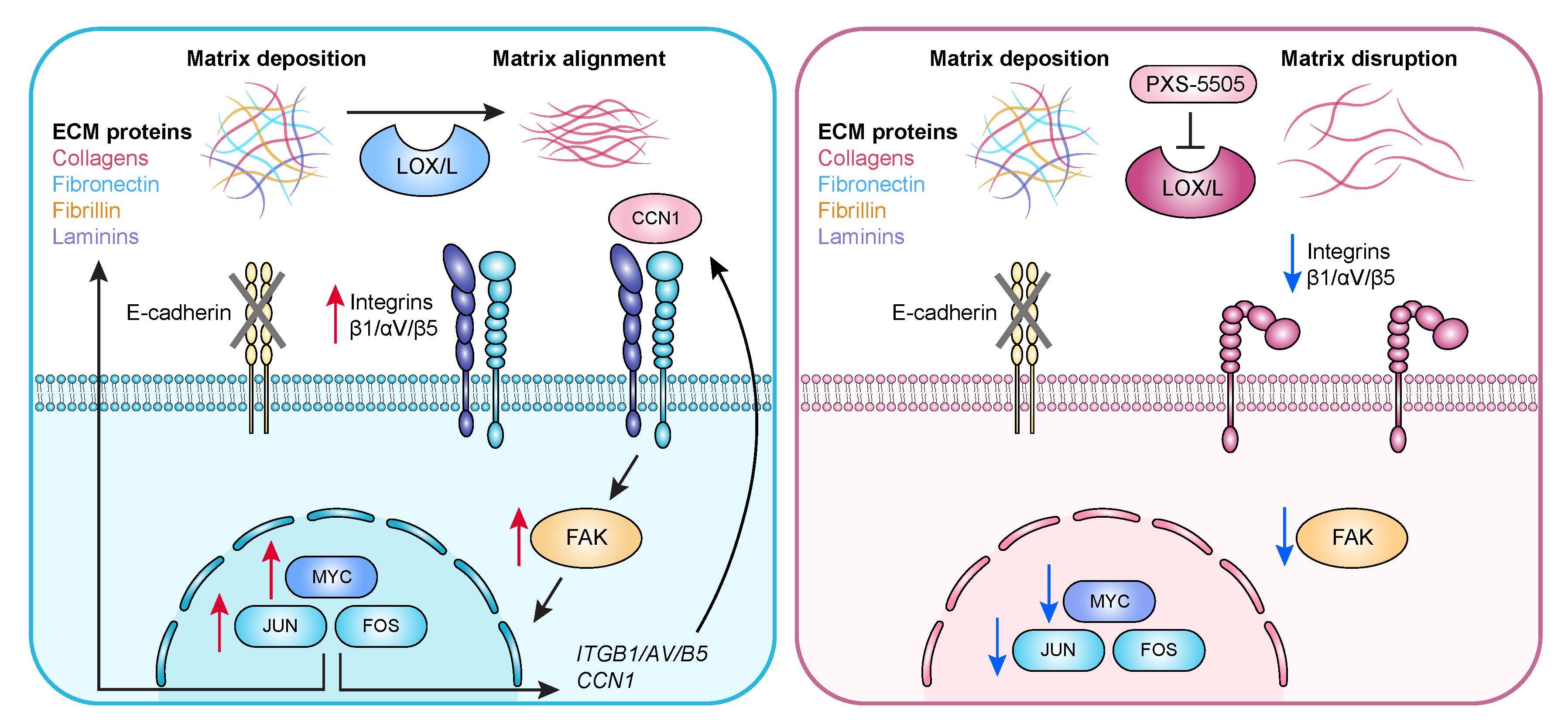
Using their advanced models, the scientists tested the drug for its effects on ILC. The drug works by blocking certain enzymes that play a critical role in the formation and maintenance of the body's connective tissues. Lobular cancers use them to crosslink collagen, a major structural component of tissue, and modify tissue stiffness.
Across several xenograft models, the drug slowed tumor outgrowth or reduced the spread of cancer cells to the lungs. It changed the structure of collagen and cancer cells lost key growth signals from their surroundings.
An actionable weakness
The team also identified measurable changes in collagen structure and gene activity that could be used to monitor whether the drug is working in short pre-surgery clinical studies (window trials).
"Our study identifies an actionable weakness in ILC-its dependence on the collagen-rich environment," says Brisken. "The drug shows potential to stop tumor growth and prevent metastasis in realistic preclinical models."
She adds: "The work provides potential biomarkers that clinicians can measure in patients to see whether the drug is effective. Most importantly, it opens the door for the first rationally designed, biology-based treatment strategy specifically for lobular carcinoma."
Other contributors
- The Institute of Cancer Research
- Tempus AI
- EPFL Bioinformatics Competence Center
- University of Lausanne
- The Francis Crick Institute
- University of Brighton
- AstraZeneca
- The Royal Marsden Hospital
- Lausanne University Hospital CHUV
- International Cancer Prevention Institute (ICPI)
- Syntara (formerly Pharmaxis)
Funding
- Breast Cancer Now Toby Robins Research Centre
- The Institute of Cancer Research
- Barts Charity
- Worldwide Cancer Research
- UK Research and Innovation (UKRI)
- China Scholarship Council program






