A research team led by Prof. YANG Weishen and Prof. ZHU Xuefeng from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) has revealed the mechanism of oxygen activation on Barium-containing perovskite materials.
The researchers discovered that BaO/BaO2 nanoparticles precipitated on the surface of Ba-containing materials under high-temperature oxygen-rich conditions had an ultra-high activity for oxygen activation, which clarified the mechanism of high-temperature oxygen activation and transport on the surface of Ba-containing perovskite oxides.
This study was published in Science Advances on April 13.
In 2000, the DICP team invented an oxygen-permeable membrane material named Ba0.5Sr0.5Co0.8Fe0.2O3-δ (BSCF). Due to its good catalytic activity towards oxygen activation, BSCF has become a representative material for oxygen permeation and has been widely used in solid oxide fuel cells, oxygen reduction reactions, and oxygen evolution reactions.
However, the essence of the good performance of the BSCF perovskite is still unclear.
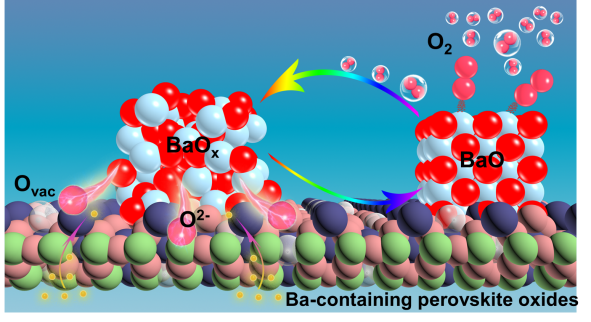
In this study, the researchers analyzed the oxygen permeation process and found that the addition of Ba into perovskite oxides could accelerate the oxygen surface exchange reaction kinetics.
They identified the precipitation of BaOx nanoparticles on the surface of BSCF materials in a high-temperature oxygen atmosphere by environmental electron microscopy, and proved that Ba-containing materials could be precipitated or decomposed into BaOx have high catalytic activity for the oxygen activation process.
Moreover, combined with DFT calculation, they found that the precipitated BaOx nanoparticles could reduce the energy barriers of oxygen molecule adsorption and dissociation in the oxygen reduction and oxygen desorption of the oxygen evolution process, thus accelerating the oxygen exchange reaction kinetics at the gas-solid interface.
"This study provides a scientific basis for the design of oxygen-permeable membranes and electrocatalytic materials," said Prof. YANG.
The above work was supported by the Pilot Project of CAS, the National Natural Science Foundation of China, and the China Postdoctoral Foundation.






