The classical protein secretion pathway considers that the secreted protein can be recognized by the signal recognition granule after the free ribosome synthesizes the signal peptide, and the ribosome is carried to the endoplasmic reticulum by the signal recognition granule to continue the protein synthesis. At the same time, under the guidance of the signal peptide, the newly synthesized protein enters the endoplasmic reticulum cavity, and is successively transported to the Golgi apparatus and plasma membrane in the form of vesicles, thereby being released. As a secreted protein transforming growth factor-β (TGF-β) cytokine, it is mainly released into the extracellular space through the above-mentioned classical protein secretion pathway to exert biological functions. IL-1β is the only cytokine known to be regulated by proteasomal degradation through ubiquitination. However, it is still unclear whether there is an intracellular regulatory mechanism of TGF-β.
On June 14, 2022, a research paper titled "Ursodeoxycholic acid reduces antitumor immunosuppression by inducing CHIP-mediated TGF-β degradation" was published online in Nature Communications by Prof. CAI Zhijian and Prof. WANG Jianli's research group from the Institute of Immunology, Zhejiang University School of Medicine. The degradation mechanism of TGF-β cytokines at the post-translational level was revealed, subverting the classical cognition.
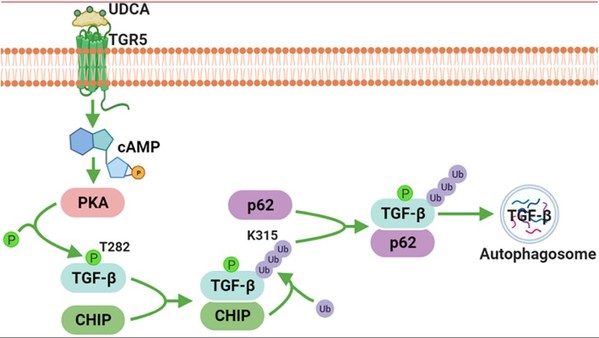
TGF-β is essential for inducing systemic tumor immunosuppression. In the latest study, researchers found that the clinically approved compound ursodeoxycholic acid (UDCA), by degrading TGF-β, enhances antitumor immunity through restraining Treg cell differentiation and activation in tumor-bearing mice. Furthermore, UDCA synergizes with anti-PD-1 to enhance antitumor immunity and tumor-specific immune memory in tumor bearing mice. UDCA phosphorylates TGF-β at T282 site via TGR5-cAMP-PKA axis, causing increased binding of TGF-β to carboxyl terminus of Hsc70-interacting protein (CHIP). Then, CHIP ubiquitinates TGF-β at the K315 site, initiating p62-dependent autophagic sorting and subsequent degradation of TGF-β. Notably, results of retrospective analysis shows that combination therapy with anti-PD-1 or anti-PD-L1 and UDCA has better efficacy in tumor patients than anti-PD-1 or anti-PD-L1 alone. Just as Prof. Cai says, "Our work demonstrates that TGF-β has an intracellular regulatory mechanism through autophagy-lysosomal degradation; besides, we also find that ursodeoxycholic acid is a ready-made TGF-β signaling inhibitor, which has extremely high application value in clinical tumor therapy.
Source: School of Medicine, Zhejiang University






