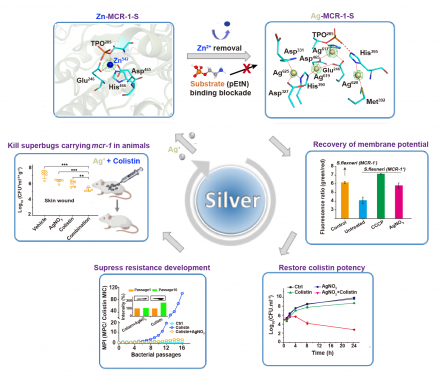
Image 1: Illustration of silver to re-sensitise colistin against pathogen carrying mcr gene in vitro and in vivo. Cartoon of active-site pocket of MCR-1-S enzyme with cofactor Zn2+ shown as the blue sphere (upper left) and the novel tetra-nuclear silver center in the active-site pocket of Ag-MCR-1-S (upper right). Ag+ shown as wheat spheres are bridged in a narrow pocket by either coordination bonds (red solid lines) or hydrogen bond (red dash line).The substitution of Zn2+ from MCR-1-S by Ag+ and inhibition of substrate (phosphorylethanolamine, pEtN) bound to MCR-1-S enzyme result in the loss of enzyme activity, which is reflected by the recovered membrane potential (middle right). Growth curves of MCR-1-J53, a pathogen carrying mcr-1 gene, treated with different drugs in vitro (lower right) and bacterial load in abscess area on day 7 in the skin of Balb/c mice infected with mcr-1 positive K. pneumoniae 9607 and received single dose of vehicle, mono-therapy of colistin, AgNO3, or their combination (i.e. AgNO3+colistin) (middle left). Resistance acquisition curves of MCR-1-J53 during 16 passages with the sub-inhibitory concentrations of colistin alone or combination of colistin and AgNO3 (lower left). The inset shows the normalized expression level of MCR-1 in MCR-1-J53 receiving different treatments.
Drug-resistant superbugs pose a huge threat to human health. Currently, colistin is regarded as the last-line antimicrobial against extensively drug resistant (XDR) bacterial infections caused by pathogens, such as Salmonella, Pseudomonas aeruginosa, Klebsiella pneumoniae, Escherichia coli, Shigella and Acinetobacter baumannii. Unfortunately, the efficacy of colistin has been seriously compromised in the regular treatment of lethal bacterial infections, owing to the emergence of mobilized colistin resistance gene-1 (mcr-1) firstly identified in 2015.
To cope with the serious challenge in the post-antibiotic era, scientists try to re-sensitise the resisted antibiotic against pathogens by using combination therapy between antibiotic and antibiotic- resistance breaker (ARB) to overcome antibiotics-resistance, which will open a window to treat these clinical infections caused by multidrug-resistant pathogens.
A research team led by Professor Hongzhe SUN, Chair Professor from the Department of Chemistry, Faculty of Science, in collaboration with Dr Pak-Leung HO, Director of the HKU Carol Yu Centre for Infection from the Department of Microbiology, Li Ka Shing Faculty of Medicine, The University of Hong Kong (HKU), discovered that an ancient antimicrobials drug, i.e., silver nitrate (AgNO3), can re-sensitise colistin against these multidrug resistant superbugs. Silver nitrate has a long antimicrobial history (more than 2,000 years) and is used to be applied in these external bacterial infections, such as skin infection.
The ground-breaking findings on antimicrobial resistance (AMR) are now published in a leading multidisciplinary science journal Proceedings of the National Academy of Sciences of the United States of America (PNAS).
The study is an extension of their past studies on antibiotics-resistance (Note 1). The research team previously discovered an anti-rheumatic agent, auranofin, as a dual-inhibitor to re-sensitise colistin and β-lactam antibiotics, of which the findings were published in Nature Communications in 2020.
"Considering the well-recorded safety history of silver nitrate as ancient antimicrobial drugs applied in external infection, silver nitrate can work with auranofin to reinforce each another in treating the infections caused by mcr-positive multidrug resistant superbugs, which provides a new hope to overcome antibiotics-resistance crisis in the post-antibiotic era," said Professor Sun.
Background
Antimicrobial resistance (AMR) occurs when bacteria mutate or adjust in response to the misuse or overuse of antimicrobial. As a result, antimicrobial cannot kill bacteria or inhibit its growth any more.
AMR has become one of the biggest public health challenges in this era. According to a report from World Health Organization (WHO), by 2021, the number of people worldwide suffering from antibiotics-resistance will be 9% more than the previous year. Indeed, it also predicted that the total number of people infected by multidrug resistant superbugs will up to tens of millions by 2050, which may even exceed the number of people suffering from cancer.
Under the special situations of the current pandemic, antimicrobial resistance caused by improper use of antibiotics has a worsening trend. The Hong Kong's Department of Health has pointed out that a study found that up to 71% of COVID-19 patients had received antibiotic treatment, and only 4% of them had bacterial infection (Note 2).
Colistin, a polymyxin E family antimicrobial, is regarded as the last-line antimicrobial against extensively drug resistant (XDR) gram-negative bacteria infections. Unfortunately, scientists have first identified the emergence of mobilised colistin resistance gene-1 (mcr-1) in 2015, which has seriously compromised in the efficacy of colistin in regular treatment of lethal bacterial infections. Worse still, owing to the rapid evolution rate of mcr, other family members have also been identified, and each has its own sub-types. These plasmid-borne resistances can cross-spread rapidly via horizontal gene transfer between bacterial strains and species, resulting in emergence of multiple drug resistant superbugs. As said by Dr Tedros Adhanom Ghebreyesus, Director-General of WHO, "As we gather more evidence, we see more clearly and more worryingly how fast we are losing critically important antimicrobial medicines all over the world." Thus, in clinical practice, common infections with these "superbugs" may soon be untreatable, which will severely endanger the public health system and leave patients with virtually no therapeutic options.
Key findings
The research team found that silver (Ag+) including silver nanoparticles can restore colistin efficacy against mcr-positive bacteria. By using X-ray crystallography method (a X-ray to observe the distribution of electron density in crystal), they confirmed that silver inhibited the activity of MCR-1 enzyme via substitution of Zn2+ (a key factor to result in colistin resistance) in the active site as well as forming a tetra-silver cluster in the active-site pocket of MCR-1. The tetra-silver center effectively inhibits the activity of MCR-1 enzyme, leading to the dysfunction of MCR-1.
The study further showed that silver effectively slows down the development of higher-level resistance and reduces mutation frequency. In the mouse-skin infection model, the combination therapy composed of AgNO3 and colistin is also highly effective in relieving the dermonecrotic lesions and reducing the bacterial load. In conclusion, the great combination effect in vitro and in vivo undoubtedly offers a promising strategy to overcome antimicrobial resistance in post-antibiotic era.
About the research team
The research was conducted by a team led by Professor Hongzhe SUN, Norman & Cecilia Yip Professor in Bioinorganic Chemistry, The University of Hong Kong. Drs. Qi ZHANG and Runming WANG from the same affiliation are co-first authors. Other HKU scientists contributing to the research included Dr Chunjiao LIU, Postdoctoral fellow (Department of Microbiology, Li Ka Shing Faculty of Medicine), Dr Mohamad Koohi-Moghadam, Research Assistant Professor (Faculty of Dentistry), Dr Haibo WANG, Postdoctoral fellow (Department of Chemistry, Faculty of Science), Dr Pak-Leung HO, Director of the HKU Carol Yu Centre for Infection (Department of Microbiology, Li Ka Shing Faculty of Medicine ), and Dr Hongyan LI, Research Assistant Professor (Department of Chemistry and State Key Laboratory of Synthetic Chemistry, Faculty of Science). Dr Minji WANG, Associate Professor from Department of Chemistry and Molecular Engineering, East China Normal University, has also contributed a lot. The work was supported by Research Grants Council of Hong Kong, Health and Medical Research Fund and Seed Fund for Basic Research and The University of Hong Kong.
About Professor Hongzhe SUN
Professor Hongzhe SUN is a Norman & Cecilia Yip Professor in Bioinorganic Chemistry and Chair Professor in the Department of Chemistry at The University of Hong Kong. His research focuses on metalloproteins, medicine chemistry (discovery of antimicrobial agents), and inorganic chemical biology. Professor Sun has published a series of well-recognized work in overcoming antimicrobial resistance against superbugs, developing anti-coronavirus agent, and understanding the functions of metalloproteins.






