The thinnest material, graphene, is the most promising materials for many applications, such as in electronics, energy storage and etc. For high performance electronic applications, synthesizing graphene directly on an insulating substrate is highly desired. To date, a great deal of efforts has been dedicated to synthesizing graphene on numerous insulating substrates, such as SiO2, Al2O3, Si3N4, BN. It is found that the growth of graphene on these insulating substrates are generally 10,000 times slower than that on metallic substrates (Figure 1) and, as a consequence, the graphene synthesized on an insulating substrate is generally with very low quality and the productivity is very low. To understand why graphene growth on an insulating substrate is so different from that on metallic substrates is critical for optimizing the experiential design but such a question has never been answered satisfactory.
 Figure 1. Graphene growth on an (left) an insulating substrate and (right) a metal substrate.
Figure 1. Graphene growth on an (left) an insulating substrate and (right) a metal substrate.
Recently, Distinguished Professor Feng Ding and his colleagues from the Center for Multidimensional Carbon Materials (CMCM), within the Institute for Basic Science (IBS) at UNIST have unveiled the mechanism of graphene growth on insulating substrates for the first time. With the state of the art theoretically computational method, they have showed that the key difference between graphene growth on insulating substrates and metallic substrate is that the state of the carbon precursor used to growth graphene. During graphene growth on a metallic substrate, a large amount of carbon precursors bond on the metal surface and can diffuse rapidly to feed the graphene growth, while, on an insulating substrate, no precursors can be trapped by the substrates and thus the graphene growth is fed by the gas phase active precursors. Through theoretical modeling, they have found that the CH3 molecules in the gas phase are the key precursors for the graphene growth and they play two important roles in graphene growth-(i) the attachment of CH3 molecules to the growth front of a graphene and (ii) remove excessive H atoms of the graphene edge via a reaction of CH3 + H CH4 (Figure 2) to ensure cleanness of the graphene edge during growth. The authors call the new mechanism of graphene growth on insulating substrate as "vapor-solid (VS) growth", which is drastically different from that mechanism of graphene growth on metallic substrates, the vapor-surface-solid (VSS) growth.
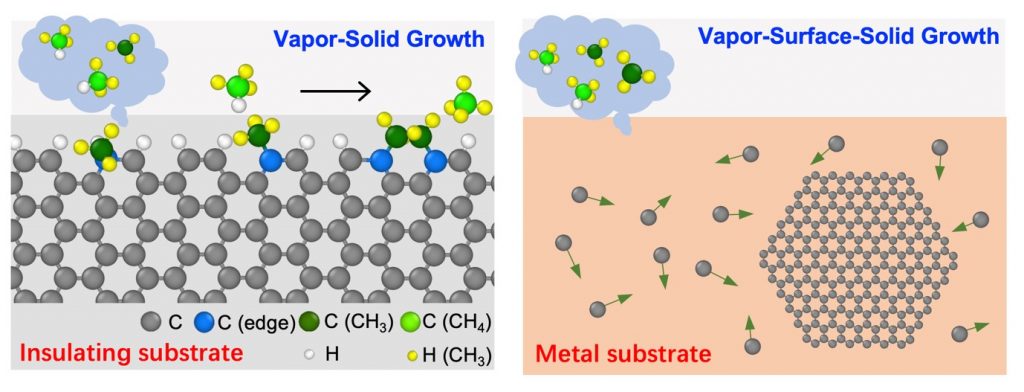
Figure 2. Schematic illustration of graphene vapor-solid growth on insulating substrates (left) and vapor-surface-solid growth on metal substrates (right).
Based on the new theory, the authors have successfully explained the very slow graphene growth on various insulating substrates. "Our calculations showed that the removal of hydrogen atoms from the graphene edge is the hardest reaction during graphene growth, plus the very low concentration of CH3 in the gas phase, the growth rate of graphene on any insulating substrate is extremely low." Ting Cheng, the first author said. "This study clearly showed the limitations of graphene growth on insulating substrates and the deep understanding on the growth mechanism leads a few potential methods to promote the graphene growth on insulating substrates, such as by using a higher growth temperature or more active carbon sources and, if possible, using more active insulating substrates to bind the precursor molecules to the substrate." Prof. Ding added.
The findings of this research have been published in the online version of ACS Nano on March 22, 2021. This work has been jointly participated by Professor Zhongfan Liu and Professor Zhirong Liu (College of Chemistry and Molecular Engineering, Peking University). This study has been supported by the Institute for Basic Science (IBS), South Korea.
Feng Ding
Distinguished Professor, School of Materials Science and Eng., UNIST
IBS Center for Multidimensional Carbon Materials (CMCM)
Dahee Carol Kim
IBS Communications Team
T: +82-42-878-8133
Story Source
Materials provided by the Institute of Basic Science.






