Acute-on-chronic liver failure (ACLF) develops in patients with chronic liver diseases, regardless of the presence of cirrhosis and represents the common final pathway leading to death. The short-term mortality rate amounts to 50-90% due to the fact that the cause of hepatocyte injury and the pathway involved in ACLF progression remain elusive. The chronic liver failure (CLIF) Consortium Acute-On-Chronic Liver Failure in Cirrhosis (CANONIC) study indicated that an intense systemic inflammatory response is the main cause of acute deterioration in patients with alcoholic liver disease-related and hepatitis C virus-related ACLF. However, there is a paucity of systematic elaboration concerning the pathogenesis of hepatitis B virus-related ACLF (HBV-ACLF) among Chinese patients.
HBV-ACLF exhibited clinical characteristics different from alcoholic liver disease-related ACLF in western populations, so the research team led by Prof. LI Jun from the First Affiliated Hospital of the Zhejiang University School of Medicine engaged in a large prospective multicenter study together with 13 hepatitis centers in China and established COSSH-ACLF criteria for the diagnosis of ACLF in the HBV-positive Chinese population. These criteria exhibited improved diagnostic sensitivity and prognostic accuracy
On this basis, researchers selected 340 patients with HBV aetiology from the COSSH study open cohort to evaluate and characterize the evolving disease course starting from chronic hepatitis B (CHB) or liver cirrhosis (LC) and progressing through acute-on-chronic hepatic dysfunction (ACHD) to ACLF and enrolled 60 healthy volunteers as normal controls. A total of 65 subjects (ACLF, 20; ACHD, 10; LC, 10; CHB, 10; NC, 15) underwent mRNA sequencing using peripheral blood mononuclear cells (PBMCs). This study identified immune-metabolism disorder triggered by HBV exacerbation as a potential mechanism of HBV-ACLF. Their research findings were published in the journal Gut.
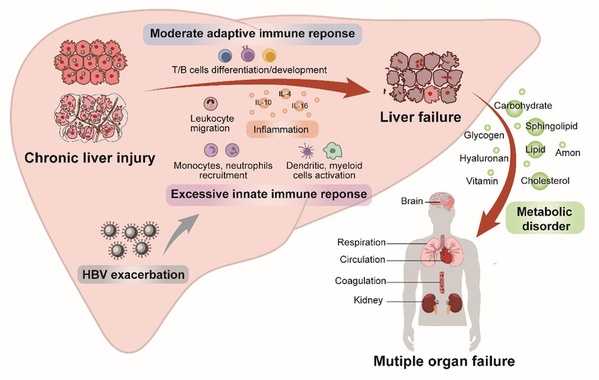
This study reveals the transcriptomic-based molecular mechanism underlying disease development. To be more specific, immune-metabolism disorder is identified as a core axis of disease development and progression in patients with HBV-ACLF. Meanwhile, four differentially expressed genes (PPAR gamma, MER tyrosine kinase, thrombospondin 1 and semaphorin 6B) are identified as potential biomarkers that reflect dysregulated pathways underlying the virus-based immune-metabolism disorder in patients with ACLF.
"The study may provide directions for the development of effective early diagnostic and treatment strategies targeting immune regulation and metabolic balance to reduce the high mortality rate of patients with HBV-ACLF," said LI Jun.






