The research team led by LIU Jianxiang from the Zhejiang University College of Life Sciences, recently published an article entitled "XBAT31 regulates thermoresponsive hypocotyl growth through mediating degradation of the thermosensor ELF3 in Arabidopsis" in Science Advances, an top-tier open access multidisciplinary journal published by American Association for the Advancement of Science's (AAAS) This study reveals that XBAT31 functions as a major E3 ligase to mediate ELF3 degradation in response to warm temperature, representing a new regulatory layer of complexity in temperature signaling during plant thermomorphogenesis.
Temperature is a major factor governing the seasonal behavior and distribution of plants on earth. Understanding the molecular mechanisms underlying plant responses to elevated ambient temperature is particularly relevant in the context of global climate change, and importantly, for breeding thermo-tolerant crop varieties in a rational fashion. In the model plant Arabidopsis, the warm temperature alters its growth, including hypocotyl elongation, leaf hyponasty, and accelerates flowering, in a process called thermomorphogenesis. PHYTOCHROME-INTERACTING FACTOR4 (PIF4) is a key regulator of plant thermomorphogenesis, which is subjected to various regulations, both at transcriptional and posttranslational levels. The evening complex (EC), consisting of EARLY FLOWERING3 (ELF3), ELF4, and LUX ARRYTHMO (LUX), is a core component of the circadian clock and coordinates environmental temperature cues with endogenous developmental signals for thermoresponsive gene expression and hypocotyl growth. ELF3 recruits ELF4 and LUX to repress the transcriptional expression of PIF4 during early night. ELF3 also suppresses PIF4 protein activity in an EC-independent manner. Recently, the polyQ repeats of ELF3 within a predicted prion-like domain (PrD) were reported to function as a thermosensor in Arabidopsis. However, how the protein stability of ELF3 is regulated at warm temperature remains elusive.
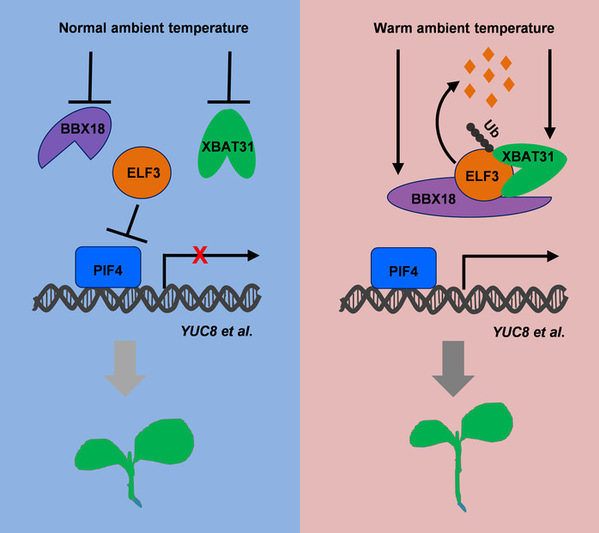
In the current study, RNA-sequencing data indicates that XBAT31, a gene encoding a putative E3 Ub ligase, is induced by warm temperature. Hypocotyl phenotyping experiments as well as genetic analysis of XBAT31 mutants confirm that XBAT31 is a novel positive regulator in thermomorphogenesis. XBAT31 interacts with ELF3, both in vitro and in vivo. Ubiquitination of ELF3 by XBAT31 in vitro demonstrates that XBAT31 can ubiquitinate ELF3 directly. Meanwhile, in vitro cell-free degradation experiments show that XBAT31 accelerates ELF3 degradation in a manner dependent on the 26S proteasome. In vivo protein immunoblotting experiments reveal that mutation of XBAT31 results in enhanced accumulation of ELF3 and reduced hypocotyl elongation at warm temperature. Interestingly, this study shows that XBAT31 interacts with the previous identified B-box protein BBX18, and the XBAT31-mediated ELF3 degradation is partially dependent on BBX18. Therefore, under warm temperature conditions, XBAT31, recruited by BBX18, interacts with ELF3, ubiquitinates ELF3, and promotes ELF3 degradation via the 26S proteasome, thereby releasing the inhibitory effect of ELF3 on PIF4 and promoting downstream gene expression for plant hypocotyl elongation.






