A research team led by Kobe University Graduate School of Medicine's Professor OGAWA Wataru (the Division of Diabetes and Endocrinology) and Project Associate Professor NOGAMI Munenobu (the Department of Radiology) has discovered that metformin, the most widely prescribed anti-diabetic drug, causes sugar to be excreted in the stool.
Metformin has been used for more than 60 years, and is the most frequently prescribed drug for diabetes in the world. Administration of metformin lowers blood sugar levels, but the mechanism behind this effect is not clear. Metformin's mode of action has thus been actively researched over the world.
Taking advantage of the new bio-imaging apparatus PET-MRI, the research team revealed that metformin promotes the excretion of blood sugar from the large intestine into the stool. This is a completely new discovery that has never previously been predicted.
The current finding may explain metformin's biological actions for which the underlying mechanism is unknown, and contribute to the development of new drugs for diabetes.
These findings were published on June 3, 2020 in the online edition of Diabetes Care, a medical journal published by the American Diabetes Association.
Main points
- Metformin is the most frequently prescribed drug for diabetes in the world. The mechanism by which this drug lowers blood sugar concentration is not clear.
- In a bioimaging study on humans, metformin was found to promote the "excretion of sugar into the stool".
- This newly discovered action of metformin may explain some of the drug's biological effects and contribute to the development of new medication for diabetes.
Research Background
Diabetes is characterized by the elevation of blood sugar concentration, which damages the blood vessels and in turn leads to various diseases. More than 400 million people suffer from diabetes around the world, therefore the prevention of diabetes and its related diseases is an important global medical issue.
A number of drugs that reduce blood sugar concentration are available. Among them, metformin is one of the oldest classes of drugs and has been used for more than 60 years. Metformin, recommended as a first-line drug in many countries, is the most frequently prescribed medication for diabetes.
However, the mechanism by which metformin lowers blood sugar concentration is not clear. Elucidation of this mechanism would contribute to the development of new and better drugs for diabetes. Consequently, research has been actively conducted into the action of metformin.
Summary of the Discovery
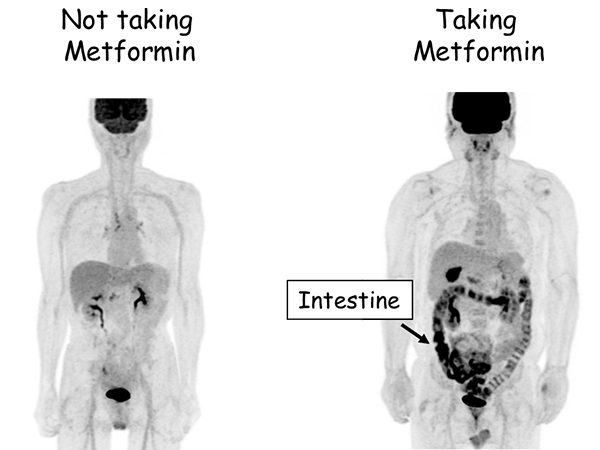
Figure 1. Representative FDG-PET MRI images of patients taking and not taking metformin.
The areas where FDG (sugar) is accumulated appear black. In patients taking metformin (right), the intestine appears black, which indicates that FDG (sugar) is accumulated in the intestine.
FDG-PET (fluorodeoxyglucose-positron emission tomography) is an imaging test to study where and how much FDG (a substance similar to sugar) is accumulated in the body after the administration of this substance through the vessels. Because FDG behaves in a similar way to sugar in the human body, FDG-PET can reveal organs or tissues that consume or accumulate large amounts of sugar (*1).
FDG-PET is generally conduced with a device that integrates both a PET and a CT (computed tomography) device. Obtaining images using FDG-PET and CT allow for the detailed examination of locations where FDG is accumulated. Recently, a device in which PET and MRI (magnetic resonance imaging) are integrated (PET-MRI) has been developed. MRI is used to examine the inside of the body using a strong magnetic field. It can examine bodily structures that cannot be analyzed by CT. PET-MRI is still a rare and valuable device; for example, only 9 units have been installed in Japan.
Professor Ogawa's research team used PET-MRI to investigate the movement of sugar in the bodies of diabetic patients, both those who were taking metformin and those who were not. The team found that sugar (i.e. FDG) is heavily accumulated in the intestine of patients taking metformin (Fig. 1). To understand where in the intestine sugar accumulates, the research team subsequently investigated the "wall of the intestine" and the "inside of the intestine (stool and other contents)" separately using a special technique.
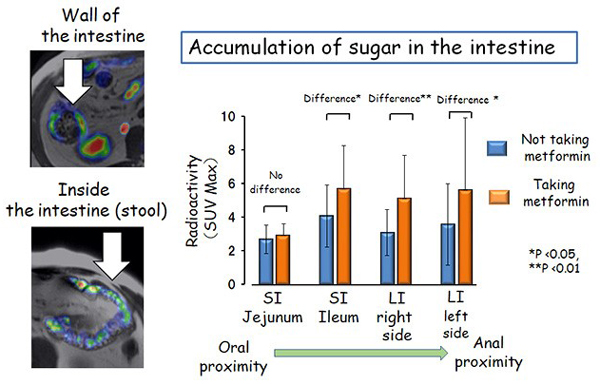
Figure 2 Taking metformin promotes sugar accumulation inside the intestine.
Left: Representative images of FDG accumulation in the intestinal wall (above) and inside (below) of the intestine.
Right: Taking metformin results in a larger amount of sugar accumulating inside the intestine distal on the anal side of the small intestine (ileum). SI, small intestine, LI, large intestine.
They found that, in patients taking metformin, more sugar was accumulated in the areas inside the intestine that are distal to the ileum (the anal side part of the small intestine) (Fig. 2). On the other hand, there was no difference in sugar accumulation in the "wall of the intestine" between patients who were taking and not taking metformin.
These results indicate that, when a patient takes metformin, sugar in the blood is released from the intestine into the stool. Not only the finding that metformin promotes the excretion of sugar into the stool, but also the revelation that sugar is excreted from the intestine into the stool itself are new discoveries that were not anticipated.
Recently, a new antidiabetic drug has been launched; the SGLT2 inhibitor, which lowers blood sugar concentrations by excreting sugar in the urine. Its beneficial clinical effects are attracting much attention. Excreting sugar into the stool, as triggered by metformin, may also be related to the SGLT2 inhibitor's glucose lowering effect.
The significance of this research and its future development
Previous studies using PET-CT showed that FDG was accumulated in the intestines of patients taking metformin. It was however assumed that FDG (sugar) was accumulated in the "wall of the intestine" without sufficient evidence because PET-CT cannot separately show the wall and the inside the intestine. In the current study, the new imaging technology PET-MRI allowed the research team to investigate the accumulation in the wall and the inside of the intestine (stool) separately, revealing for the first time that metformin-induced accumulation of sugar occurred exclusively inside the intestine.
Taking a SGLT2 inhibitor results in the excretion of tens of grams of sugar per day in the urine. In this study, it was not possible to quantitatively evaluate how many grams of sugar are excreted in the stool. The significance of this discovery will be further confirmed by using a new imaging method that will enable the excreted sugar in the stool to be quantified.
It is thought that changes in the intestinal flora caused by metformin (*2) are related to its blood sugar lowering effect, but how metformin alters the intestinal flora is completely unknown. Since changes in nutrients such as glucose affect the growth of bacteria, it is possible that metformin's effect of excreting glucose into the intestine may also be related to the changes in the intestinal flora.
Glossary
- ※1
- Since cancers consume more sugar than normal tissue, FDG-PET is generally used to find cancers that are difficult to detect using other tests.
- ※2
- The human intestine is inhabited by 100 to 1000 trillion intestinal bacteria, and these bacteria as a whole are referred to as intestinal flora. It is known that changes in the intestinal flora are related to the pathogenesis of diseases and the effects of drugs.
Journal Information
- Title
- "Enhanced Release of Glucose into the Intraluminal Space of the Intestine Associated with Metformin Treatment as Revealed by [18F]Fluorodeoxyglucose PET-MRI"
- Authours
- Yasuko Morita1, Munenobu Nogami2, Kazuhiko Sakaguchi1, Yuko Okada1, Yushi Hirota1, Kenji Sugawara1, Yoshikazu Tamori1,3, Feibi Zeng2, Takamichi Murakami2, and Wataru Ogawa1*
- 1Division of Diabetes and Endocrinology, Department of Internal Medicine; 2Department of Radiology; and 3Division of Creative Health Promotion, Department of Social/Community Medicine and Health Science, Kobe University Graduate School of Medicine, Kobe, Japan
* Corresponding author
- Journal
- Diabetes Care






