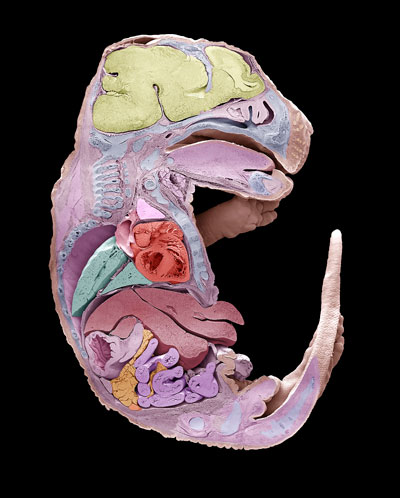
 Figure 1: A scanning electron micrograph of one half of a mouse embryo. The other half would be different in some respects. RIKEN biologists have discovered the role that genes play in the development of this left-right asymmetry. © STEVE GSCHMEISSNER/SCIENCE PHOTO LIBRARY
Figure 1: A scanning electron micrograph of one half of a mouse embryo. The other half would be different in some respects. RIKEN biologists have discovered the role that genes play in the development of this left-right asymmetry. © STEVE GSCHMEISSNER/SCIENCE PHOTO LIBRARY
A better knowledge of the causes of disease, birth defects and genetic syndromes could come from new insights gleaned by RIKEN biologists into how mice embryos develop asymmetry between their left and right sides1.
Embryos start off as symmetric bundles of cells, but then eventually develop into animals whose left sides differ from their right sides. Developmental biologists want to discover the origins of this left-right asymmetry since such knowledge will shed light on the basic biology of development, as well as on the causes of birth defects and genetic syndromes.
The first step in establishing left-right asymmetry in an embryo is the left-right breaking event. In fish, frog and mouse embryos, this begins with hair-like cilia generating fluid flow that runs leftwards. This fluid flow then downregulates Dand5 mRNA on the left-hand side of the embryo.
Now, Hiroshi Hamada of the RIKEN Center for Developmental Biology and his team, together with researchers in Switzerland and elsewhere in Japan, have investigated the factors that suppress Dand5 mRNA.
They first tracked down the critical portion of the mRNA by comparing sequences between mammalian genes to uncover the conserved portion. This revealed a conserved 200-nucleotide region of the proximal 3′-UTR.
 Hiroshi Hamada (left), Katsura Minegishi (right) and co-workers have investigated the mechanism behind the creation of left-right asymmetry in vertebrate development. © 2021 RIKEN
Hiroshi Hamada (left), Katsura Minegishi (right) and co-workers have investigated the mechanism behind the creation of left-right asymmetry in vertebrate development. © 2021 RIKEN
Embryos lacking a functional copy of the Bicc1 gene exhibited defects in left-right patterning, linking the Bicc1 protein with left-right asymmetry. When the researchers deleted the first exon of Bicc1 using CRISPR-Cas9 gene editing, the resulting mouse embryos developed symmetrically.
The team discovered that the Bicc1 protein binds to the GACGUGAC sequence in the untranslated region of Dand5 mRNA. Further work showed that Bicc1 also needed to interact with the Cnot3 component of the Ccr4-Not deadenylase complex.
These findings significantly advance knowledge of the development of left-right asymmetry. "Our study has revealed how cells respond to fluid flow generated by cilia," says Hamada. "We have confirmed that left-right symmetry is broken by degradation of a particular mRNA on the left side in response to the directional fluid flow. Discovering the involvement of the RNA degrading system in sensing the fluid flow is an exciting breakthrough."
Since Bicc1 has been implicated in kidney disease, Hamada thinks a similar mechanism may operate when cells sense other types of fluid flows, such as kidney epithelial cells detecting urine flow.
The team next wants to see how calcium activates the Bicc1-Ccr4 complex, and, in particular, whether calcium affects complex formation or phosphorylation of Bicc1 and Ccr4 components.






