Credit: Dani Bergy & Inessa Stanishevskaya/Cognition Studio, Inc.
All the cells of the body come from the same tiny clump of stem cells and share the same set of genes. But by following different patterns of activating those genes, they quickly embark towards different destinies, becoming tissues as diverse as liver, blood and brain.
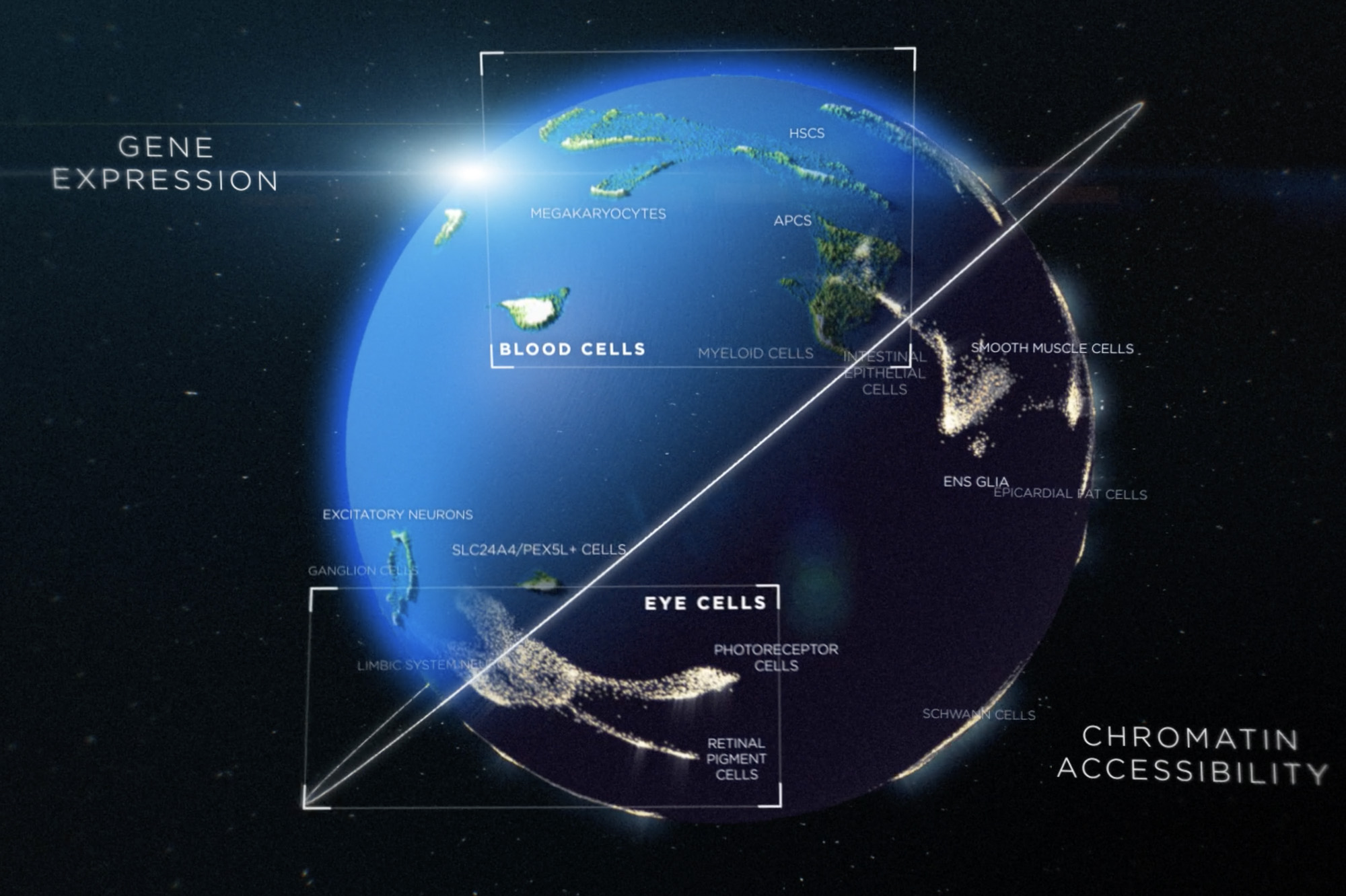
Tracking this process in humans-who consist of 37 trillion cells, many hundreds of cell types, and roughly 20,000 genes-has long been an elusive goal. Now, using an innovative technique, researchers have created an atlas providing a global view of the diverse trajectories that human cells undergo in forming different organs. Unprecedented in its scale, it allows scientists to look up gene expression profiles in singles cell from all the main organs, during different stages of development. The findings are published in Science.
"From these data, we can directly identify all major cell types across human tissues and track the patterns of gene expressions that got them there," says Junyue Cao, who completed this work in the laboratory of Jay Shendure at the University of Washington School of Medicine before joining Rockefeller this year as an assistant professor and head of the Laboratory of Single-cell Genomics and Population Dynamics.
Profiling millions of cells at once
Conventional methods allow scientists to study active genes in a mixed population of cells, or of just a few thousand individual cells at a time. Cao, however, has developed a technique that can profile over two million individual cells at once, without even having to physically separate them.
In this technique, called sci-RNA-seq3, researchers pour a hodgepodge of cells obtained from different tissues into hundreds of tiny wells containing molecular tags that attach to different molecules inside the cells. They then mix the cells and pour them again, until all molecules of the cells are tagged, and each cell ends up with its own unique combination of tags-a barcode that corresponds to its distinctive collection of molecules.
By reading this barcode, scientists can identify which genes are active in a cell, and group them into types and subtypes using machine-learning algorithms.
In creating the new atlas, the researchers applied this technique on over 4 million human fetal cells from 15 organs. They identified 77 main cell types and 657 cell subtypes, some of which had not been characterized previously-likely because they are too rare to be picked up by conventional techniques.
The data turned up several surprises that Cao hopes to investigate further in his new lab. For example, the researchers found certain embryonic cells and precursors of liver cells circulating in unexpected tissues. They also found red blood cell production in the adrenal gland, a site of hormone production. "I think there's actually quite a lot of new biology we can learn," he says.
Moreover, the researchers can now get clues about how a cell decides to specialize into a brain cell, blood cell, or any other cell type. For example, by combining the new dataset with one that they previously created of mouse embryonic cells, they can identify the genes that are evolutionarily conserved between the two species and regulate the molecular programs underlying the development of each cell type.
A second study by Cao and his University of Washington colleagues, also published in Science, provides further insight into the factors that regulate gene expression. There, the researchers used a combination of methods to create an atlas of the accessibility of chromatin, the material that tightly packs the DNA and like a control switch, determines which individual genes can be read.
Beyond providing insight into early development, the findings can help study developmental diseases, and the techniques can be used to study the maintenance of cells throughout life and its disruption in cancer and aging-related disorders.






