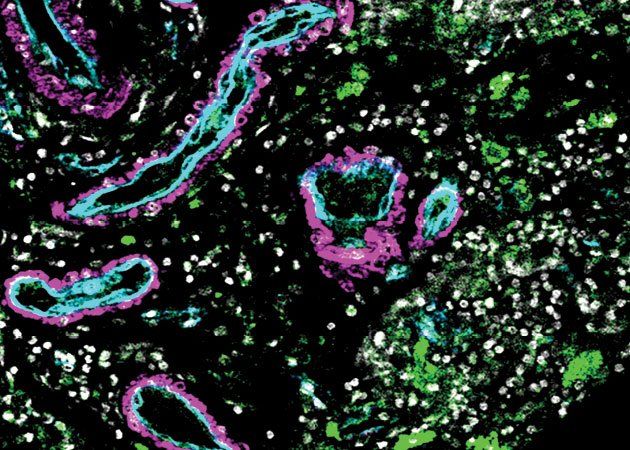
In work that will enhance the study of such disparate diseases as stroke and dementia, researchers at UC San Francisco have catalogued all the cells that form the blood vessels of the human brain, along with their locations and the genes transcribed in each.
The atlas characterizes more than 40 previously unknown cell types, including a population of immune cells whose communication with the brain's vascular cells contributes to the bleeding of a hemorrhagic stroke. This devastating form of stroke accounts for 10-15 percent of all strokes in the U.S., mostly among younger people. About half of hemorrhagic strokes are fatal.
The findings will serve as a foundation for new research on brain vasculature globally, the scientists said.
"This research gives us the map and the list of targets to start developing new therapies that could change the way we treat a lot of cerebrovascular diseases," said Ethan Winkler, MD, PhD, a neurosurgeon and research associate at the UCSF Weill Institute for Neurosciences and one of the lead authors of the study, which appears in the Jan. 27 issue of Science.
Tangles in the Brain's Vasculature
The team, headed by Adib Abla, MD, associate professor of Neurological Surgery and Daniel Lim, MD, PhD, professor of Neurological Surgery, both members of the UCSF Weill Institute for Neuroscience, along with Tomasz Nowakowski, PhD, analyzed cells in arteriovenous malformations, or AVMs, tangles of poorly formed arteries in the brain that are often the source of a hemorrhagic stroke. They compared the AVMs with samples of normal brain vasculature from five volunteers who were already undergoing brain surgery for epilepsy.
UCSF, ranked #1 in neurosurgery by U.S. News and World Report, is a leading national center for brain AVM surgery and care.
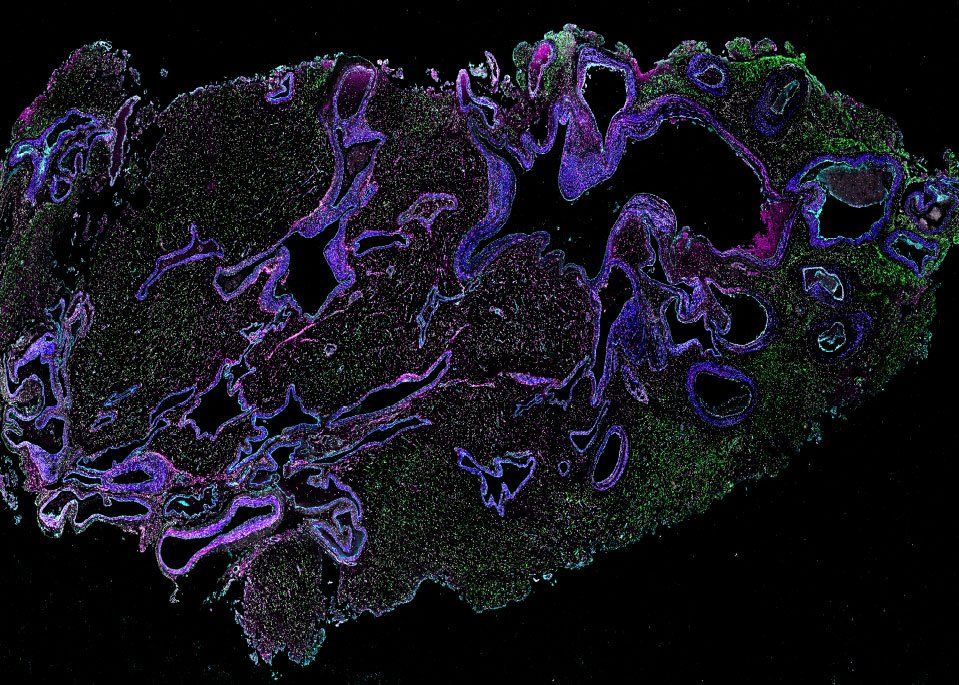
Some of the 44 samples of AVM tissue, acquired during delicate surgeries performed by Abla, Chief of Neurological Surgery, had been removed from the patient's brain while still intact, and other samples were only removed after they had started to bleed. The three varieties of tissue - normal, intact AVMs, and AVMs that had bled - allowed the researchers to get a fuller picture of differences between how the cells function normally and in different states of disease.
In collaboration with the Cerebrovascular Research Center, the team used single-cell mRNA sequencing on more than 180,000 cells to determine which genes were being expressed in the differing samples and matched gene expression with a cell's location. Chang Kim, a graduate student in bioinformatics at UCSF and co-lead author of the study, then developed computer analyses that compared gene expression in the normal and diseased cells.
An Immune Cell Surprise
The results revealed not only a variety of new cell types, but a population of immune cells that appear to communicate with smooth muscle cells in the diseased arteries and weaken them, resulting in a stroke. Scientists have suspected that the immune system is activated by malformations like AVMs. But Nowakowski said, "without this study, we wouldn't be able to pinpoint this very specific population of cells in the blood that might be the key drivers of disease progression."
Identifying these specific immune cells completely changes how researchers can think about treating this sort of vascular disease, he added. If the cells circulate in the blood, it may be possible to reduce stroke risk by modulating the immune system.
"This opens up huge therapeutic potential," said Nowakowski.
Many forms of dementia, including Alzheimer's, appear to have a vascular underpinning. We need an atlas like this to better understand how changes in the vasculature can contribute to the loss of cognition and memory.
That potential extends beyond stroke. The map can help investigate any neurovascular disease, including one of the most common: dementia.
"Many forms of dementia, including Alzheimer's, appear to have a vascular underpinning," said Lim. "We need an atlas like this to better understand how changes in the vasculature can contribute to the loss of cognition and memory."
"This work was a really a beautiful collaboration between surgeon-scientists and molecular biologists, occurring in a place with incredible access to clinical specimens," said Lim. "It's what makes the Weill Institute of Neuroscience at UCSF so special."
While many institutions don't have access to all of these critical resources, they will have access to the dataset from this study, Lim added. Nowakowski believes that this information will allow researchers across the world to perform much less expensive analyses on large numbers of patients, which is the only way to get a fuller picture of how vascular diseases operate.
"Understanding cerebrovascular disease at the cellular and molecular level will take the work of many researchers into new directions," Lim said.
A 'Periodic Table of Cell Types'
The team's study contributes to the Human Cell Atlas, an international effort to create cell reference maps for the entire body.
Nowakowski calls these atlases a "periodic table of cell types." Just as the chemical periodic table organizes elements into a structure that allows chemists to draw relationships between them based on where they appear in the table, human cell atlases reveal the locations of cells in the body and the resulting interactions between them.
While there is a lot of work taking place around the world to generate these atlases for different organs and tissues, many of them only map the geographic locations of cells. The comparison of normal and abnormal cells in this research takes it to a higher level, providing extremely refined guidance for drug development.
"Our study really demonstrates how a cell atlas can be utilized," Nowakowski said. "With our 'periodic table' as a reference, we can start asking which cells might go wrong in disease and very precisely target those cells for therapy."
Authors: Additional authors include Jayden Ross, Joseph Garcia, Eugene Gil, David Wu, Kunal Raygor, Kazim Narsinh, Helen Kim, Shantel Weinsheimer, Daniel Cooke, Nalin Gupta, and Edward Chang of UCSF. For other authors, please see the study.
Funding: This work was supported by NIH grants U54NS065705, R01NS034949, R01NS099268, R01EB012031, R01NS034467, 5P01AG052350, R01NS112357, F32CA228372 and U01MH115747, a Brain Aneurysm Foundation grant, Schmidt Futures AI Accelerator grant, and other philanthropy.
The University of California, San Francisco (UCSF) is exclusively focused on the health sciences and is dedicated to promoting health worldwide through advanced biomedical research, graduate-level education in the life sciences and health professions, and excellence in patient care. UCSF Health, which serves as UCSF's primary academic medical center, includes top-ranked specialty hospitals and other clinical programs, and has affiliations throughout the Bay Area.






