Rice is one of the most important crops in the world, particularly in China. The rice yield is most severely hindered by Magnaporthe oryzae. According to the National Agro-Tech Extension and Service Center, approximately 400 acres of rice will be plagued by rice blast in 2022. Therefore, it is of supreme importance to reveal the pathogenic mechanism of Magnaporthe oryzae and discover potential pesticide targets for prevention and control of rice blast.
The research team led by TAO Zeng from Zhejiang University College of Agriculture and Biotechnology and KOU Yanjun from China National Rice Research Institute published an open-access article entitled "The additional PRC2 subunit and Sin3 histone deacetylase complex are required for the normal distribution of H3K27me3 occupancy and transcriptional silencing in Magnaporthe oryzae" in the journal New Phytologist on July 16. This study reveals how Polycomb Repressive Complex 2 (PRC2) regulates normal distribution of facultative heterochromatin and the stable maintenance of gene repression, and provides a more insightful understanding of the pathogenic mechanism of Magnaporthe oryzae, thereby offering theoretical guidance for the prevention and control of rice blast.
Development in higher organisms requires proper gene silencing, partially achieved through trimethylation of lysine 27 on histone H3 (H3K27me3). The core subunits Kmt6-Suz12-Eed are indispensable for H3K27me3-mediated transcriptional silencing in Magnaporthe oryzae. Furthermore, these core subunits play an essential role in fungal development, stress response and pathogenicity to the host. However, the regulation on the normal distribution of H3K27me3 occupancy remains unclear, especially in fungi. Besides, Polycomb repressive complex 1 (PRC1) mediates transcriptional repression through H3K27me3 recognition, chromatin compaction and H2AK119 monoubiquitylation, but it remains unknown as to how PRC2 performs stable transcriptional silencing of target genes in the absence of PRC1 in the fungi.
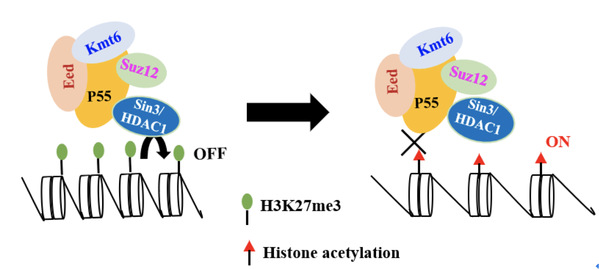
In this study, the researchers identified P55 as an additional PRC2 subunit and explored its critical role in the normal distribution of H3K27me3 and transcriptional silencing. They also found that loss of p55 could cause severe global defects in the normal distribution of H3K27me3 and transcriptional reprogramming on the H3K27me3-occupied genes. Furthermore, Sin3 histone deacetylase complex was found to be essential for sustaining H3K27me3 occupancy and stably maintain gene repression by directly interacting with P55.
To recap, this study revealed a novel mechanism by which P55 and Sin3 engaged in the normal distribution of facultative heterochromatic modifications and the stable maintenance of gene repression in eukaryotes.






