Abstract
A research team, affiliated with UNIST has demonstrated that liquid electrolytes, when frozen, can still facilitate lithium-ion conduction sufficient for battery operation-challenging the traditional view that electrolytes must be in liquid form to function. The study also revealed the mechanism of lithium-ion transfer in organic ice electrolytes, opening new possibilities for developing solid-like electrolytes for lithium metal batteries (LMBs).
Led by Professor Hyun-Kon Song from the School of Energy and Chemical Engineering at UNIST and Professor Dong-Hwa Seo from the Department of Materials Science and Engineering at KAIST, the team created an organic ice electrolyte based on ethylene carbonate (EC), a cyclic carbonate commonly used in commercial batteries. They examined how well it conducts ions and how effectively it transports lithium ions in its frozen state. Their results showed that these electrolytes operate through a hopping mechanism within a solid structure formed by immobilized solvent molecules.
In conventional Li-ion batteries, the electrolyte is a liquid solution of lithium salts dissolved in organic solvents, allowing lithium ions to move between electrodes during charging and discharging. Ethylene carbonate has a melting point of around 37°C, so it is solid at room temperature (~25°C). Usually, it's mixed with other solvents to lower this melting point. However, this study designed an electrolyte with only a small amount of lithium salt, keeping it in a frozen, ice-like state.
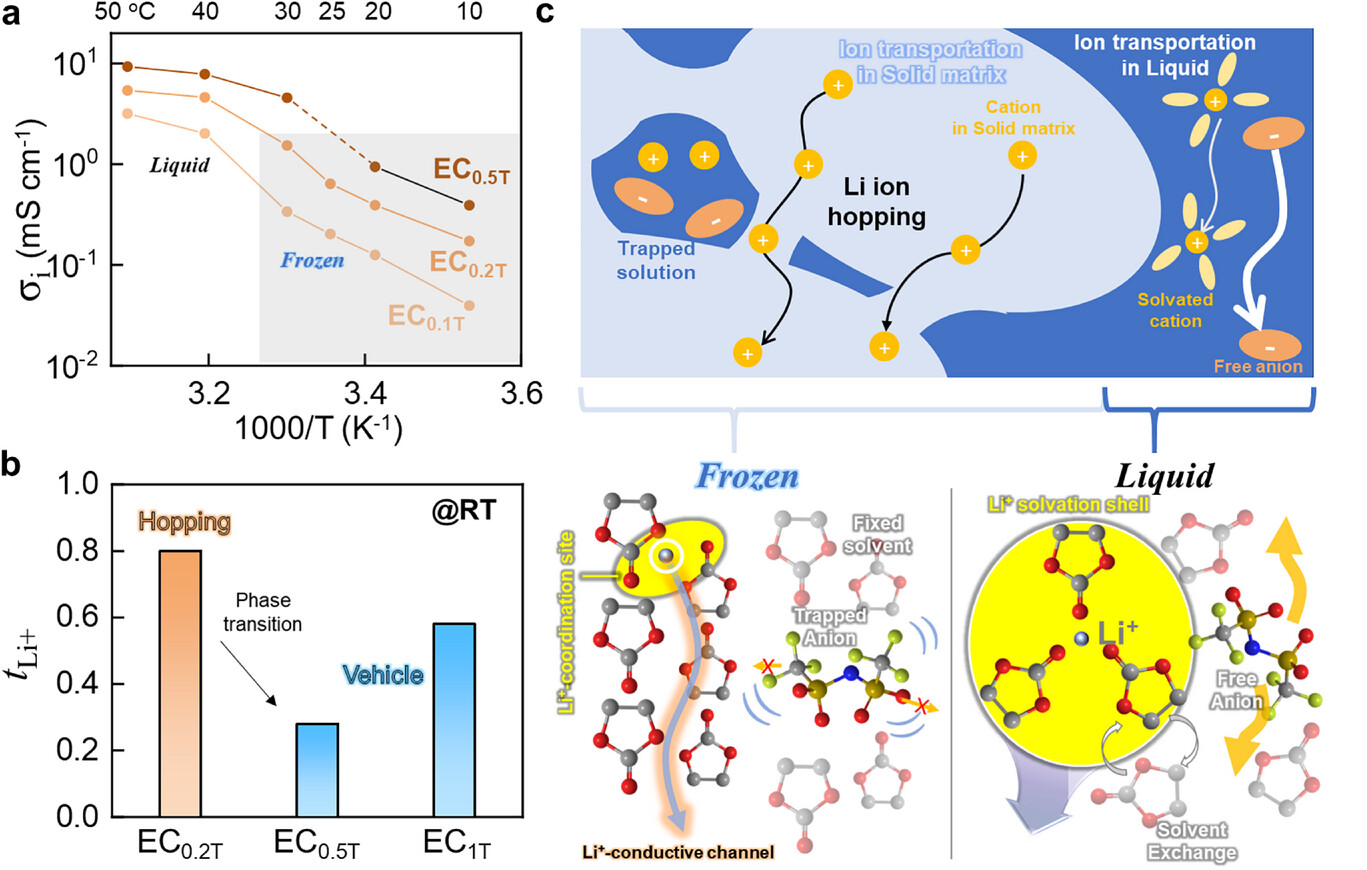 Figure 1. Ion transporting properties of organic ice electrolytes.
Figure 1. Ion transporting properties of organic ice electrolytes.
Experimental results showed that this 'ice electrolyte' had an ionic conductivity of about 0.64 mS/cm and a lithium-ion transference number of around 0.8-values comparable to advanced solid electrolytes. When used in lithium metal batteries, the frozen electrolyte supported over 400 charge-discharge cycles at room temperature without internal short circuits, demonstrating stable and reliable performance. Lithium metal batteries are seen as a promising future energy storage solution because they can store up to 50% more energy than conventional batteries. However, their wider use has been limited by issues like dendrite formation-sharp lithium deposits that can cause short circuits-and reactions between lithium metal and liquid electrolytes.
The researchers explained why the ice electrolyte performs so well. Their analysis showed that in the frozen state, solvent molecules are immobilized, and lithium ions move by hopping between neighboring oxygen atoms on the solvent molecules. This targeted ion movement reduces side reactions and prevents dendrite formation-sharp deposits that can puncture separators and cause failures.
Professor Song commented, "While many assume that solid electrolytes need to be rigid inorganic materials or special polymers, our findings show that even a loosely arranged, ice-like structure of solvent molecules can support efficient ion conduction. We are now exploring organic solvent combinations with higher melting points to make this approach practical for real-world applications."
The findings of this research have been published online in the journal Advanced Materials on January 21, 2026. The study was supported by the Samsung Research Funding & Incubation Center of Samsung Electronics and the InnoCore program of hydro*studio at UNIST.
Journal Reference
Do Sol Cheong, Minjun Kwon, Pil-Su Jung, et al., "Lithium-Ion Conduction Through Frozen Phase of Organic Electrolytes for Lithium Batteries," Adv. Mater., (2026).






