Scientists looking for the causes of neurodegenerative diseases like Parkinson's and Alzheimer's generally focus on the buildup of aberrant proteins in the brain that impede normal neural connections.
New research from Binghamton University and Drexel University looks at a different, lesser-studied issue that also hurts patients and their quality of life: how Parkinson's affects the human vascular system.
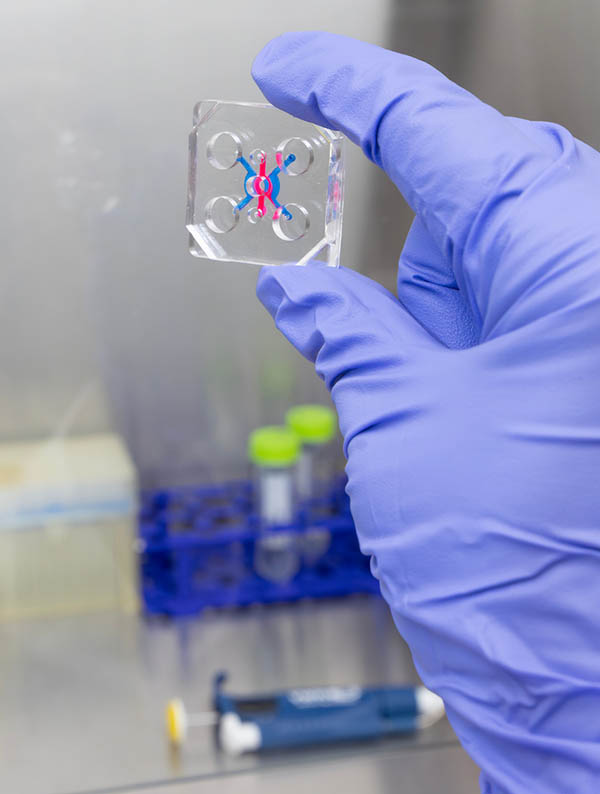
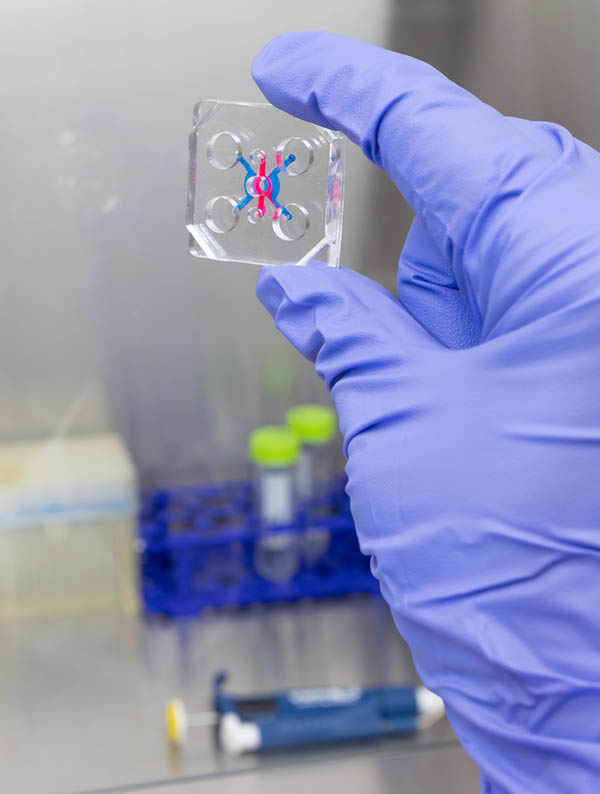
Assistant Professor Jungwook "Jay" Paek and PhD student Anika Alim from the Thomas J. Watson College of Engineering and Applied Science's Department of Electrical and Computer Engineering used organ-on-a-chip technology to mimic how cells behave inside the human body.
In 2024, Paek shared a $414,000 award from the National Institutes of Health to study how protein aggregates called Lewy bodies interact with the brain's diverse cellular environments and how it relates to the development of neurodegenerative diseases.
For this latest paper published in the Nature journal Communications Engineering, Paek, Alim, Drexel Assistant Professor Myungwoon Lee, and PhD student Yoongyeong Baek take an engineering approach to examine how the blood-brain barrier degrades as Parkinson's progresses.
"The inside of blood vessels in the brain is covered by endothelial cells that form a protective barrier to prevent harmful substances from entering the brain tissue," Paek said. "Parkinson's disease impairs the endothelial barrier functions and causes blood-vessel degeneration. Potentially, that increases neuronal exposure to circulating pathogenic substances and hurts their function as blood vessels for providing nutrients and oxygen for neurons."
Organ-on-a-chip technology cultivates living human cells in 3D microfluidic platforms about the size of a computer memory stick, and it can simulate the physiological functions, mechanics, and response of entire organs in a way that's easy to observe.
After introducing alpha-synuclein protein aggregates - similar to Lewy bodies in Parkinson's patients - into the organ-on-a-chip environment, the Binghamton and Drexel researchers tracked the progression of vascular degradation. They observed abnormalities such as endothelial dysfunction, barrier disruption, vascular regression, and impairment of blood flow.
"Before this research, most studies on Parkinson's disease have focused on the aggregation of specific proteins and their impacts on neurodegeneration," Paek said. "However, how the protein aggregation contributes to vascular complications remains largely underexplored, so this is an exciting finding."
Based on this research, future work will include more testing and improved artificial-intelligence modeling for disease progression. Paek hopes that understanding more about neurological disorders will lead to innovative therapies.
"Parkinson's is a multifaceted disease," he said. "Understanding the complex interplay between neurodegeneration and vascular pathology offers unique opportunities to help patients."
The publication, "Microengineering of the capillary interface of midbrain dopaminergic neurons to study Parkinson's disease vascular alterations," was selected for a short video feature by Springer Nature.