Urea is an extremely important chemical, especially for fertilisers. But, making urea is energy intensive and relies heavily on fossil fuels.
However, new findings from Griffith University and the Queensland University of Technology have highlighted new ways to produce urea electrochemically, using electricity and waste gases such as carbon monoxide (CO) and nitrogen oxides (NO) instead.
"The challenge is that when CO and NO react on a catalyst, they usually don't form urea," said co-lead author Professor Qin Li from Griffith University.
"Instead, they tend to make unwanted by‑products such as ammonia or hydrocarbon compounds.
"This makes selective urea production very difficult."
What did the researchers do?
The research team combined quantum chemistry simulations and machine learning to reveal better catalyst designs that encouraged CO and NO to combine and form a carbon-nitrogen bond, instead of producing unwanted side reactions.
They studied:
- Pairs of metal atoms anchored on the edges of carbon materials (called dual‑atom catalysts)
- How these metal pairs interacted with CO and NO at the same time
- Why some metals encouraged urea formation while others did not
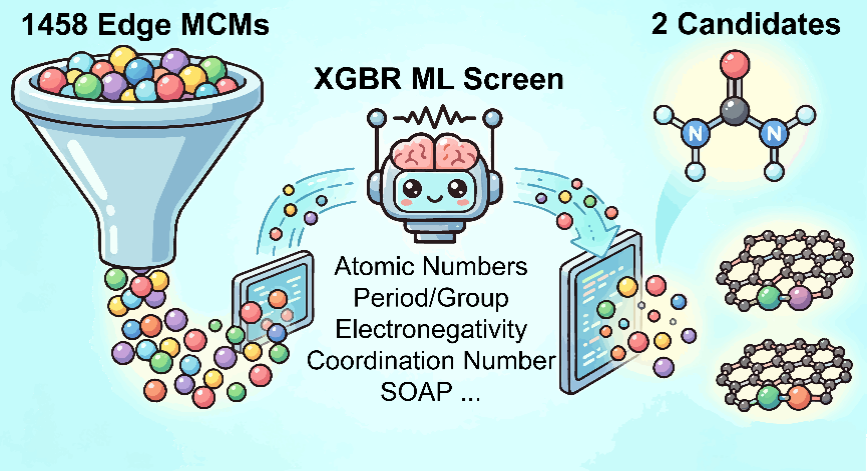
This resulted in the examination of 90 catalyst designs using high‑accuracy computer simulations, then using machine learning to rapidly screen more than 1,400 additional candidates.
What was the key outcome?
The most important discovery was how strongly CO and NO stuck to the catalyst together, not how each gas stuck on its own.
The team identified a single number, called the "co‑adsorption energy", that reliably predicted whether a catalyst would make urea, or instead make ammonia or hydrocarbons.
"We found a very narrow 'sweet spot' for this energy," co-lead author Dr Yun Han said.
"If CO and NO bound too weakly, they fell off the surface.
"If they bound too strongly, the gases got over‑reduced and formed the unwanted side products.
"Only moderate binding strength favoured urea formation."
Why was machine learning important here?
The research team said testing thousands of catalyst designs with physics‑based simulations would take years.
To shorten that timeframe, the team developed a machine-learning model using simple atomic properties (from the periodic table) and structural information about the carbon edges.
The model accurately predicted the key co‑adsorption energy and allowed the researchers to narrow 1,458 possible catalysts down to 259 promising ones, then validate only the best few with simulations.
"This approach dramatically accelerates catalyst discovery," said co-lead author, computational chemist Professor Aijun Du from Queensland University of Technology.
"This study provides a clear design rule for making urea catalysts, and shows how machine learning and chemistry can solve complex reaction problems. This moves urea production closer to a low‑carbon, sustainable process, and offers a reusable blueprint for designing catalysts for other green chemical reactions.
Professor Aijun Du
"We can systematically design catalysts that turn waste gases into fertiliser efficiently, rather than relying on lengthy and costly trial and error."
The paper 'Machine Learning-Assisted Design Framework of Carbon Edge-Dominated Dual-Atom Catalysts for Urea Electrosynthesis' has been published in ASC Nano.










