A team of researchers, affiliated with UNIST has introduced a novel responsive system, capable of molecular recognition of physicochemically similar mixtures, such as isotopes. This will provide a simple, cost-effective solution for generating heavy hydrogen, which are raw materials essential for the nuclear fusion power generation.
Professor Hoi Ri Moon and her research team, in collaboration with Professor Hyunchul Oh from Gyeongnam National University of Science and Technology (GNTECH) and Professor In Hwan Oh from Korea Atomic Energy Research Institute (KAERI) have unveiled a new phenomenon, in which heavy hydrogen (i.e., deuterium) is selectively adsorbed by flexible metal-organic frameworks (MOFs) under given temperature and pressure conditions. Using this principle, the separation of deuterium from gaseous hydrogen
Using this principle, deuterium can be efficiently separated from hydrogen gas.
The research result was selected as a cover paper in the Journal of the American Chemical Society (JACS) in recognition of the importance of the research, and was released on July 14th.

176 spine minimum. full size. Editor: Lingling JEM: Leslie
RTP: Michael Reid
The "arc reactor" in the movie Iron Man uses nuclear fusion power to create explosive energy. Deuterium is the raw material of this arc reactor. It is a'isotope' of hydrogen that has more neutrons than general hydrogen. It contains only a very small amount of about 0.016% of the total hydrogen gas, making it difficult to separate and expensive. There is a method of separating deuterium using a'flexible metal-organic skeleton' that has many holes inside and the size of the holes changes, but it requires precise temperature and pressure control, and the amount that can be separated is small. This is because it is difficult to meet the conditions in which only deuterium can be 'adsorbed' in the pores.
Professor Hoe-ri Moon's research team discovered that'MIL-53′, a representative'flexible metal-organic skeleton', has a'second breathing phenomenon' that opens only in deuterium under a certain temperature and pressure. They devised a way to obtain deuterium. Both hydrogen and deuterium cause a 'breathing reaction (first breath)' that expands the pores of the flexible metal-organic skeleton like a lung, but the second breathing reaction in which the pores expand once again occurs only by deuterium. In the existing separation method, both deuterium and normal hydrogen can penetrate and pass through the pores, so the conditions for adsorbing only deuterium in the pores had to be precisely adjusted. On the other hand, if the'second breathing phenomenon' discovered this time is used, deuterium can be separated with high efficiency without any separate manipulation.
The research team conducted deuterium separation experiments using "MIL-53," a flexible metal-organic skeleton that looks like a long rubber tube with both ends open. Small pores with a size of 0.26 nm (nanometer, 1 nm = 1 billionth of a meter) inside the skeleton grow to 0.67 nm from the moment when hydrogen gas is encountered at cryogenic temperatures (-253°C), where the temperature is increased (-248°C). ) If the pressure is maintained at 800 ~ 1000 mbar, the pore expands (pore size 0.86 nm) again by the second breathing phenomenon. The second breathing phenomenon was caused only by deuterium.
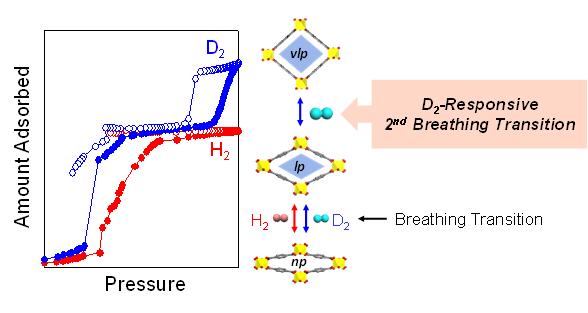
As a result of the experiment, the amount of deuterium that can be adsorbed additionally due to the second breath is 32 mg per 1 g of porous material, which is 2.5 times the amount of deuterium (12 g) isolated in the previous study. In addition, the research team further analyzed the structural change of the metal-organic skeleton caused by the selective adsorption of deuterium into the pores through neutron scattering experiments.Through this, high-density deuterium exists in the pores of MIL-53(Al) under the conditions. Showed that.
"Through this study, we were able to once again prove the potential of the flexible metal-organic framework in the separation of hydrogen isotopes," says Professor Moon. "we believe that this study opens new avenues for developing novel smart materials capable of simultaneously recognizing and responding to specific isotope molecules and for a wide range of applications."
Professor Oh Hyeon-cheol said, "This research is of great significance in discovering a new adsorption phenomenon in which the structure of the flexible metal-organic skeleton changes structure by selectively reacting only to specific isotopes." Economical isotope separation and deuterium enrichment are possible without designing and processing the separation system."
This research, which was jointly participated by Professor Kang Seong-gu of Ulsan University and Dr. Park Ji-Tae of the German Nuclear Fusion Research Institute (FRM-II), was carried out with support from the Korea Research Foundation's mid-sized researcher support project and the nuclear basic research project.
Journal Reference
Jin Yeong Kim, Jaewook Park,Junsu Ha, et al., "Specific Isotope-Responsive Breathing Transition in Flexible Metal-Organic Frameworks," J. Am. Chem. Soc, (2020).






