A research team led by Prof. WANG Junfeng from the Hefei Institutes of Physical Science (HFIPS) of the Chinese Academy of Sciences (CAS) has developed a new one-dimensional (1D) fluorine-19 (19F) nuclear magnetic resonance (NMR)-based method for rapid detection of Arf6 guanine nucleotide exchange factor (GEF) activity.
Results were published in Analytical Chemistry on June 6.
Small GTPases (sGTPase) are involved in signal cascades that control a wide range of cellular responses. All sGTPases can be found in two states: an inactive state in which the small GTPase is bound to Guanosine diphosphate (GDP), and an active state in which it is bound to Guanosine triphosphate (GTP). The GTPase cycle formed by the interconversion between the two states is strictly regulated by guanine nucleotide exchange factors (GEFs) and GTPase activating proteins (GAPs).
Mutation or abnormal regulation of sGTPase proteins or their respective GEFs/GAPs are closely related to many diseases including cancer. Therefore, it is important to detect the activity of these GEF/GAP proteins both in vitro and in vivo.
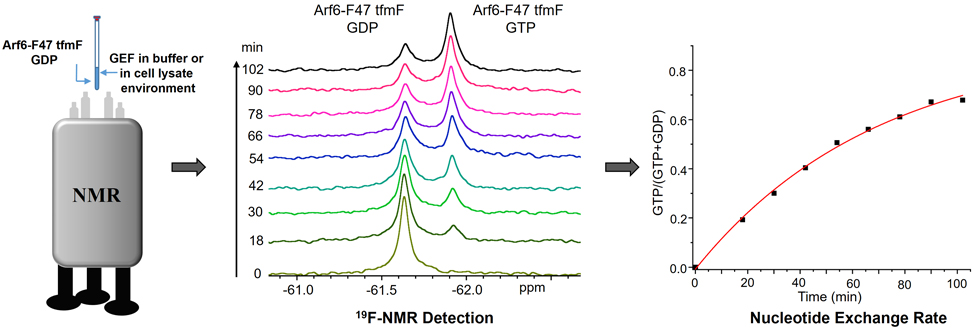
In this study, the researchers incorporated the unnatural amino acid tfmF site-specifically into Phe47 of sGTPase Arf6, a position located in the ARF6 switching domain whose conformation changes most during GTPase cycle. And they developed the 1D 19F NMR-based method to detect GEF activity in purified protein system or in the presence of the cell lysates.
For a GEF activity assay, the sample of GDP-loaded Arf6-F47 tfmF was put into the spectrometer as soon as possible after adding a GEF and GTP. A sequential series of 1D 19F NMR spectra were collected. The signal of Arf6-F47 tfmF in the GTP-bound state was processed and integrated. The exchange kinetic curves were then plotted and fitted to a single-phase exponential growth curve to extract the rate.
"This strategy could potentially be used to monitor conformational change of Arf6 or other sGTPase," said WU Bo, first author of the study, "it can detect the activities of sGTPases regulatory proteins in physiology and pathology environments."
This study was supported by the National Natural Science Foundation of China, the Collaborative Innovation Program of Hefei Science Center, and the High Magnetic Field Laboratory of Anhui Province. All the NMR experiments were performed at the High Magnetic Field Laboratory of CAS.