In a study published online in Journal of Experimental Medicine, Dr. XIAO Yichuan's group from Shanghai Institute of Nutrition and Health (SINH) of the Chinese Academy of Sciences (CAS), and their collaborators, discovered that high concentration of TGFβ inhibits the expression of BFAR in CD4+ T cells so as to impair TH9 differentiation as well as Th9-mediated tumor immunotherapy, and elucidated the underlying molecular mechanism.
Immunotherapy has become one of most promising directions in the global clinical treatment for tumor. In recent years, through promoting anti-tumor immune response, PD-1/PD-L1-blocking therapy has been used to cure several kinds of tumors. However, it is only effective in a small subset of people. Therefore, to study the mechanism of the insensitivity of this therapy is the key to improve the effect of immune checkpoint therapy.
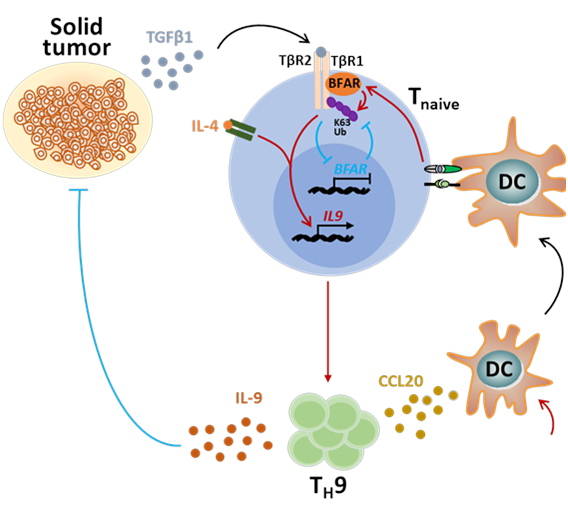
Worked as an immune suppressor, TGFβ has been reported to be highly-expressed in multiple kinds of tumor tissues and is able to regulate tumor immune escape. The blockage of TGFβ signaling pathway would enhance the effect of tumor immunotherapy because TGFβ is known to induce the differentiation of Th9 cells, a subset of CD4+ T cells with obvious anti-tumor effect. Whether TGFβ1 could promote TH9 cells differentiation and exhibit anti-tumor effect is not clear.
Unexpectedly, the researchers found that high concentration of TGFβ1 in plasma from tumor patients was inversely correlated with IL-9 levels. High level TGFβ1 can negatively regulate the activation of TGFβ signaling pathway and TH9 cell inducibility by inhibiting the expression of BFAR, which impairs anti-tumor immune response.
Mechanistically, BFAR mediates K63-linked ubiquitination of TGFβR1 at K268, which is critical to activate TGFβ signaling. Thus, BFAR deficiency or K268R knock-in mutation suppresses TGFβR1 ubiquitination and TH9 differentiation, thereby inhibiting TH9-meidated cancer immunotherapy.
In addition, the researchers found that BFAR-overexpressed TH9 cells exhibited promising therapeutic efficacy to curtail tumor growth and metastasis in mouse cutaneous melanoma model and the lung metastasis model, and increased the sensitivity of the PD-1 antibody mediated immunotherapy. Using a patient-derived xenograft (PDX) colorectal tumor model in immunodeficient NCG mice, they found that BFAR-OE TH9 cells showed ideal effect in eliminating tumor growth.
This study discovered BFAR as a key TGFβ1-regulated gene to fine-tune TGFβ signaling activity and TH9 induction, which provides a new strategy for enhancing the therapeutic efficacy of TH9 mediated tumor immunotherapy.