Joint press release by Max-Planck-Institut für Kohlenforschung and Hokkaido University
A New study reveals that the stereochemistry of lipid nanoparticles critically influences safety and efficacy in mRNA delivery.
A team from the Max-Planck-Institut für Kohlenforschung, Hokkaido University, and Osaka University has discovered that subtle differences in molecular structure can have a major impact on the performance of mRNA-based drugs. Their findings, published in the Journal of the American Chemical Society, open the door to the development of safer and more effective vaccines and therapies.
To deliver therapeutic nucleic acids like mRNA into cells, scientists rely on lipid nanoparticles (LNPs), tiny, fat-based carriers that protect fragile genetic material, enabling it to survive in the body and reach target cells. A key component of these LNPs are ionizable lipids, which help mRNA enter cells and then release it effectively. One such lipid, ALC-315, was notably used in the Pfizer/BioNTech COVID-19 vaccine, a medical breakthrough that played a critical role in controlling the global pandemic.
The overlooked impact of stereochemistry
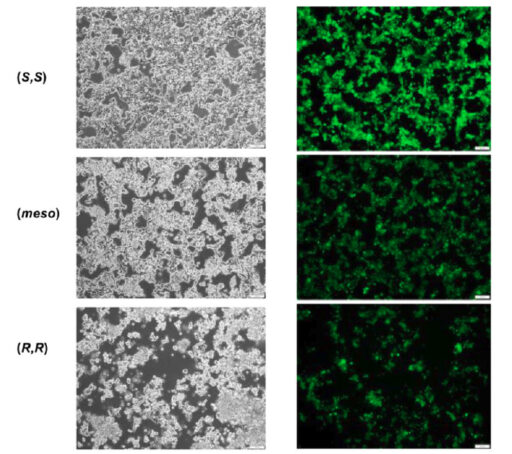
While the vaccine is safe and highly effective, one surprising detail has remained largely unnoticed: ALC-315 is used as a mixture of three stereoisomers-molecules that have the same chemical formula but differ in their three-dimensional arrangement, much like left and right hands. This distinction is not trivial. It is well known that stereoisomers can behave very differently in biological systems, affecting how drugs are absorbed, distributed, and metabolized. Until now, the three isomers of ALC-315,(S,S), (R,R), and meso, had never been individually studied in the context of LNP performance. In their groundbreaking study, Dr. Chandra Kanta De from the Max-Planck-Institut für Kohlenforschung and his colleagues synthesized and tested each pure stereoisomer for the first time. "We demonstrate that stereochemistry directly impacts both the efficacy and safety of LNPs," explains Dr. De. "In particular, we found that the (S,S)-form delivers mRNA just as efficiently as the standard mixture, but with significantly reduced toxicity." Dr. Masumi Tsuda from Hokkaido University, Japan, who conducted the biological analysis of the ALC-315 isomers with Prof. Shinya Tanaka, mentioned, "We would be delighted if these research findings could contribute to the development of safer vaccines."
These findings are highly relevant, as LNP technology is rapidly expanding beyond COVID-19 vaccines, with applications ranging from gene therapy to personalized cancer vaccines. As the field pushes toward the next generation of mRNA-based therapeutics, the need for precise, optimized components becomes increasingly important. The study paves the way for improved LNP formulations, offering the potential for safer and more effective mRNA delivery in broad spectrum of future therapies.
The work was supported by the Max Planck Society, the German Research Foundation (DFG), the European Research Council (ERC), the Japan Society for the Promotion of Science, and the Japan Agency for Medical Research and Development (AMED).
Original article:
Chandra Kanta De et al., The Overlooked Stereoisomers of the Ionizable Lipid ALC315. Journal of the American Chemical Society, July 31, 2025.
DOI: 10.1021/jacs.5c08345
Funding:
This study was supported by the Max Planck Society, the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation; Germany's Excellence Strategy, RESOLV − EXC 2033, ID: 390677874); the European Research Council (ERC, European Union's Horizon 2020 research and innovation program "C−H Acids for Organic Synthesis, CHAOS" Advanced Grant Agreement No. 694228 and European Union's Horizon 2022 research and innovation program "Early Stage Organocatalysis, ESO" Advanced Grant Agreement No. 101055472) ; JSPS KAKENHI (23K06584, 22H04989, and 24H00037); the Japan Agency for Medical Research and Development (AMED) (JP24ek0410124h, JP24gm1810006h, and 223fa627002h).






