The provenance of oxygen on Earth and other solar planetary bodies is a fundamental issue. It is widely accepted that the prebiotic pathway of oxygen production in the Earth primitive atmosphere was via vacuum ultraviolet (VUV) photodissociation of CO2 and subsequent recombination of two O atoms.
In contrast, the photodissociation of H2O, one of the dominant oxygen carriers, has long been assumed to proceed mainly to produce hydroxyl (OH)- and hydrogen (H)-atom primary products, and its contribution to oxygen production is limited.
Recently, a research group led by Prof. YUAN Kaijun and YANG Xueming from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences revealed oxygen production from the three-body photodissociation of water molecule using the Dalian Coherent Light Source.
Their findings were published in Nature Communications on April 30.
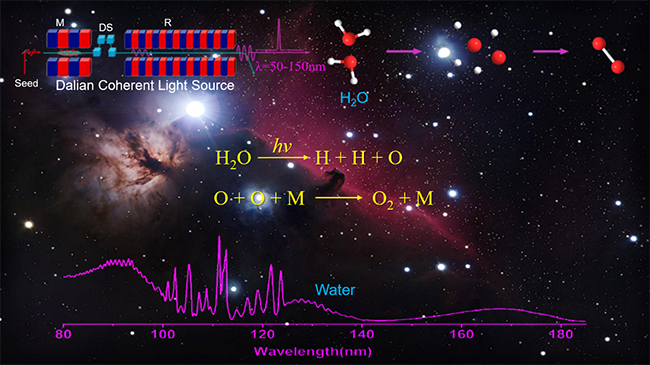
The VUV free-electron laser facility at the Dalian Coherent Light Source allows the researchers to quantitatively assess the importance of H2O photochemistry for oxygen production.
"Our experimental results indicated that H2O under VUV excitation can break into three fragments: one O atom and two H atoms, where the O atoms are in the 1D and 3P states. The three-body dissociation process is the dominant channel for H2O photochemistry in the 90-110 nm region," said Prof. YUAN.
The quantitative determination demonstrated that approximately 20% of the H2O photoexcitation events resulted in O atoms. Considering the water abundance in widely interstellar circumstances such as in interstellar clouds, atmospheres of the solar-family comets, and even in the Earth primitive atmosphere, O production from water photolysis must be an important process. The subsequent recombination of O atoms produced O2, which represented an important prebiotic O2-production pathway.
This research was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences, Chemical Dynamics Research Center, and the National Natural Science Foundation of China.






