A first-in-human trial from Institute of Science Tokyo and EVA Therapeutics, Inc., demonstrates that enteral ventilation, a technique that could deliver oxygen through the intestine, is safe and feasible in healthy subjects. The method involves intrarectal administration of an oxygen-carrying fluid into the gut, where oxygen may diffuse into the bloodstream. These findings provide important evidence for further testing of enteral ventilation as a potential alternative oxygenation method for patients with respiratory failure.
Enteral Ventilation for Oxygen Delivery via the Intestine: A First-in-Human Trial
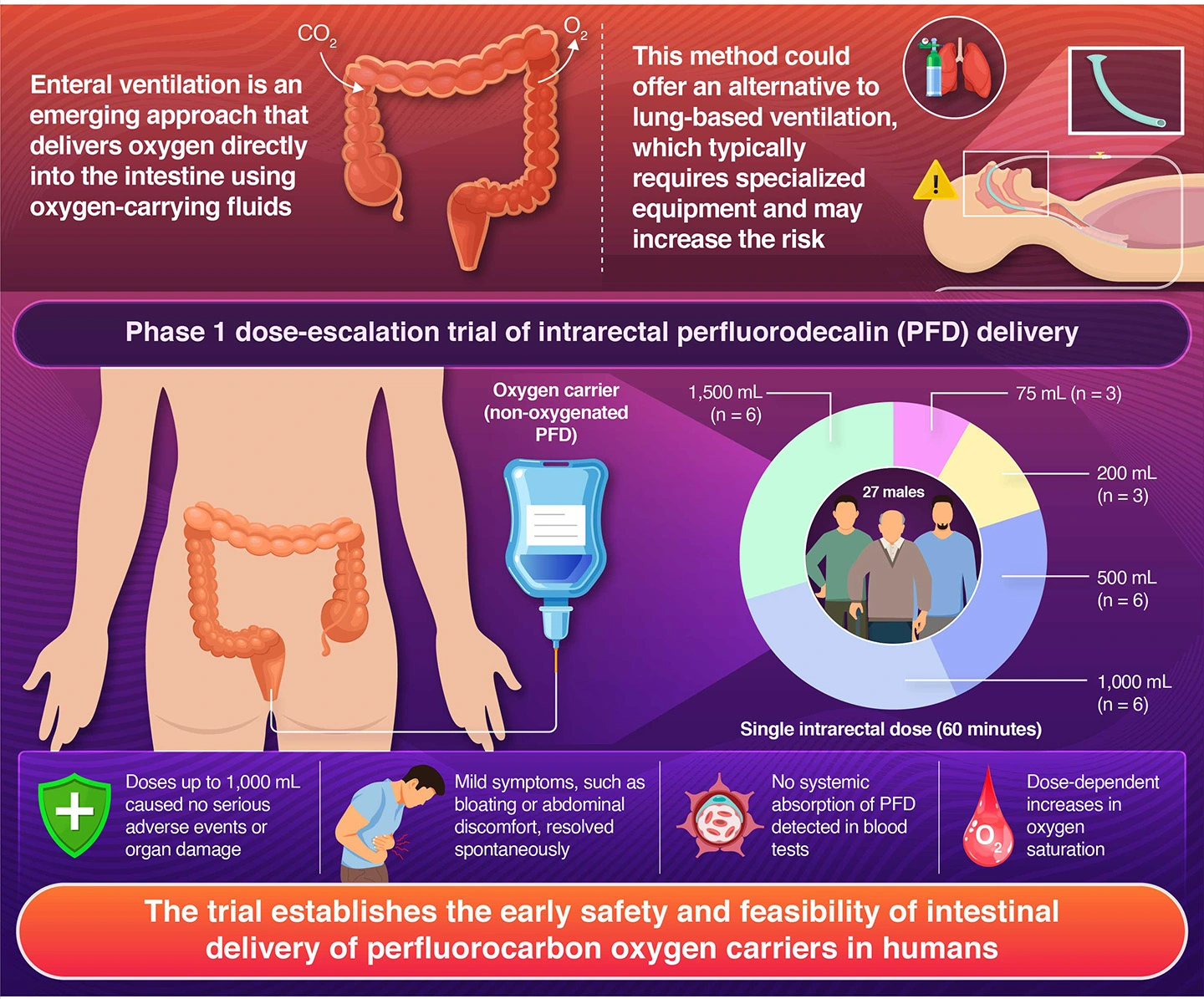
For patients suffering from respiratory failure, the standard oxygen support methods involve supplying oxygen into the lungs. However, most oxygenation strategies require specialized equipment and expertise and, in some cases, can also damage the lungs when air is delivered at very high pressure or when the lungs are overstretched by large volumes of air.
Now, a trial conducted by a research team led by Professor Takanori Takebe from the Human Biology Unit, Institute of Integrated Research, Institute of Science Tokyo (Science Tokyo), Japan, who is also a Professor at the Department of Genome Biology, the University of Osaka Graduate School of Medicine, Japan, and Deputy Director of the Premium Research Institute for Human Metaverse Medicine (WPI-PRIMe), The University of Osaka, Japan, together with Associate Professor Tasuku Fujii from the Department of Anesthesiology, Nagoya University Graduate School of Medicine, Japan, points toward the feasibility of an alternative approach in which oxygen could be delivered through the intestines. This technique, known as enteral ventilation, uses oxygen-carrying liquids called perfluorocarbons, which can potentially deliver oxygen through the intestine and into the bloodstream.
Earlier investigations in animal models showed that this method could improve oxygen levels in the body. Now, the findings of this study provide the first evidence that the procedure is generally safe and feasible in humans, paving the way for a potential new approach to treating respiratory failure.
This paper was made available online on October 20, 2025, and was published in Volume 6 Issue 12 of the journal Med on December 12, 2025.
"We confirmed the safety and tolerability of enteral ventilation. These findings provide the foundational safety data to advance enteral ventilation as a novel therapeutic strategy for patients with respiratory failure," says Takebe.
The researchers conducted a clinical trial in which 27 healthy male volunteers between the ages of 20 and 45 years received a single intrarectal dose of non-oxygenated perfluorodecalin (PFD), a perfluorocarbon liquid that can carry oxygen. The participants were given increasing amounts of PFD ranging from 25 mL to 1,500 mL. They were asked to retain the liquid in the intestine for up to 60 minutes while researchers monitored them for any adverse effects.
Participants receiving doses up to 1,000 mL generally tolerated the treatment well. At the highest dose of 1,500 mL, more than half of the participants experienced mild abdominal pain, leading some to stop the procedure early as a precaution. Overall, 20 out of 27 participants successfully retained the full dose.
The most common side effects were mild and temporary, including a sense of urgency to defecate, abdominal pain, and bloating. Notably, all symptoms resolved on their own once the participants expelled the PFD naturally. Blood tests confirmed that the PFD remained confined to the intestine and was not absorbed into the bloodstream. Researchers also observed no clinically significant changes in markers of liver or kidney function.
While these findings are promising, the researchers emphasize that the trial used non-oxygenated PFD primarily to evaluate the safety of the material and the procedure. The potential therapeutic effects of fully oxygenated perfluorocarbons will need to be examined in future studies.
"These preliminary findings support the clinical feasibility of enteral PFD delivery and lay the groundwork for its application in designing oxygen-PFD dosing strategies in future clinical studies," says Takebe.
If successful, enteral ventilation could offer a new strategy for treating severe respiratory conditions and may also help make certain medical procedures safer, particularly those in which low oxygen levels are a common complication.
Reference
- Authors:
- Tasuku Fujii1, Yasuyuki Kurihara2, Yoshihiko Tagawa3, Hirofumi Nagai2,4, Chihiro Yokota4, Hiroyuki Mizuo4, and Takanori Takebe2,5,6,7,8,9*
- Title:
- Safety and tolerability of intrarectal perfluorodecalin for enteral ventilation in a first-in-human trial
- Journal:
- Med
- Affiliations:
- 1Department of Anesthesiology, Nagoya University Graduate School of Medicine, Japan
2Institute of Integrated Research, Institute of Science Tokyo (Science Tokyo), Japan
3Nemoto Science Co. Ltd., Japan
4EVA Therapeutics, Inc., Japan
5Department of Genome Biology, the University of Osaka Graduate School of Medicine, Japan
6Division of Gastroenterology, Hepatology & Nutrition, Developmental Biology, Center for Stem Cell and Organoid Medicine (CuSTOM), Cincinnati Children's Hospital Medical Center, USA
7Department of Pediatrics, University of Cincinnati College of Medicine, USA
8Communication Design Center, Advanced Medical Research Center, Yokohama City University Graduate School of Medicine, Japan






